 | |
 | |
| Clinical data | |
|---|---|
| Pronunciation | /tɛˈstɒstəroʊn ənˈdɛkənoʊeɪt/ teh-STOS-tə-rohn ən-DEK-ə-noh-ayt |
| Trade names | Oral: Kyzatrex, Andriol, Jatenzo, others IM: Aveed, Nebido, others |
| Other names | TU; Testosterone undecylate; Testosterone 17β-undecanoate; ORG-538; CLR-610 |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a614041 |
| License data | |
| Pregnancy category |
|
| Routes of administration | By mouth, intramuscular injection |
| Drug class | Androgen; Anabolic steroid; Androgen ester |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | Oral: 3–7% Intramuscular: high |
| Protein binding | High (testosterone) |
| Metabolism | Liver |
| Metabolites | Testosterone, undecanoic acid, metabolites of testosterone |
| Elimination half-life | IM (in tea seed oil): 20.9 days[6][7] IM (in castor oil): 33.9 days[6][7] |
| Excretion | ~90% Urine, 6% feces |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| ECHA InfoCard | 100.025.193 |
| Chemical and physical data | |
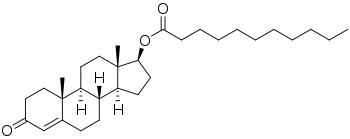
| Formula | C30H48O3 |
| Molar mass | 456.711 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Testosterone undecanoate, sold under the brand names Andriol, Aveed and Nebido among others, is an androgen and anabolic steroid (AAS) medication that is used mainly in the treatment of low testosterone levels in men,[8][6][9][10][11][12][13] It is taken by mouth or given by injection into muscle.[10][14]
Side effects of testosterone undecanoate include symptoms of masculinization like acne, increased hair growth, voice changes, hypertension, elevated liver enzymes, hypertriglyceridemia, and increased sexual desire.[10] The drug is a prodrug of testosterone, the biological ligand of the androgen receptor (AR) and hence is an androgen and anabolic steroid.[15][10] It has strong androgenic effects and moderate anabolic effects, which make it useful for producing masculinization and suitable for androgen replacement therapy.[10] Testosterone undecanoate is a testosterone ester and a prodrug of testosterone in the body.[9][8][6] Because of this, it is considered to be a natural and bioidentical form of testosterone.[16]
Testosterone undecanoate was introduced in China for use by injection and in the European Union for use by mouth in the 1970s.[17][18] It became available for use by injection in the European Union in the early to mid 2000s and in the United States in 2014.[19][20] Formulations for use by mouth are approved in the United States.[3][4][21] Along with testosterone enanthate, testosterone cypionate, and testosterone propionate, testosterone undecanoate is one of the most widely used testosterone esters.[15][6][10] However, it has advantages over other testosterone esters in that it can be taken by mouth and in that it has a far longer duration when given by injection.[22][8][6][7][10] In addition to its medical use, testosterone undecanoate is used to improve physique and performance.[10] The drug is a controlled substance in many countries and so non-medical use is generally illicit.[10]
Oral administration of testosterone undecanoate is an effective method to achieve therapeutic physiological levels of serum testosterone in patients with hypogonadism. In addition, oral therapy has been found to have a positive impact on quality of life factors such as sexual function, mood, and mental status, as documented in various studies.[23]
Medical dosage
Testosterone undecanoate is used in androgen replacement therapy in men, including trans-men. It is specifically approved only for the treatment of hypogonadism.[24][25][26] As an intramuscular injection, it is administered at a dosage of 1,000 mg (4 mL) once every 3 months.[14] Because of rare occasions of pulmonary embolism it is not recommended to inject 4 mL of Nebido into one muscle, but to split it into two 2mL injections into left and right counter muscles (e.g. gluteals).[27]
Conversely, oral testosterone undecanoate must be taken two or three times a day with food.[14][28]
Side effects
Side effects of testosterone undecanoate include virilization among others.[10]
Anaphylaxis
The Reandron 1000 formulation (Aveed in the United States) contains 1,000 mg of testosterone undecanoate suspended in 4 ml castor oil with benzyl benzoate for solubilization and as a preservative, and is administered by intramuscular injection. As an excipient in Reandron 1000, benzyl benzoate has been reported as a cause of anaphylaxis (a serious life-threatening allergic reaction) in a case in Australia.[29] Bayer includes this report in information for health professionals and recommends that physicians "should be aware of the potential for serious allergic reactions" to preparations of this type.[30] In Australia, reports to the Adverse Drug Reactions Advisory Committee (ADRAC), which evaluates reports of adverse drug reactions for the Therapeutic Goods Administration (TGA), show several reports of allergic reactions since the anaphylaxis case from 2011.
Pharmacology
Pharmacodynamics
| Medication | Ratioa |
|---|---|
| Testosterone | ~1:1 |
| Androstanolone (DHT) | ~1:1 |
| Methyltestosterone | ~1:1 |
| Methandriol | ~1:1 |
| Fluoxymesterone | 1:1–1:15 |
| Metandienone | 1:1–1:8 |
| Drostanolone | 1:3–1:4 |
| Metenolone | 1:2–1:30 |
| Oxymetholone | 1:2–1:9 |
| Oxandrolone | 1:3–1:13 |
| Stanozolol | 1:1–1:30 |
| Nandrolone | 1:3–1:16 |
| Ethylestrenol | 1:2–1:19 |
| Norethandrolone | 1:1–1:20 |
| Notes: In rodents. Footnotes: a = Ratio of androgenic to anabolic activity. Sources: See template. | |
Testosterone undecanoate is a prodrug of testosterone and is an androgen and anabolic–androgenic steroid (AAS). That is, it is an agonist of the androgen receptor (AR).
Pharmacokinetics
Testosterone undecanoate has a very long elimination half-life and mean residence time when given as a depot intramuscular injection.[31][6][7] Its elimination half-life is 20.9 days and its mean residence time is 34.9 days in tea seed oil, while its elimination half-life is 33.9 days and its mean residence time is 36.0 days in castor oil.[6][7] These values are substantially longer than those of testosterone enanthate (which, in castor oil, has values of 4.5 days and 8.5 days, respectively).[31] Testosterone undecanoate is administered via intramuscular injection once every three months or so.[14][32]
Chemistry
Testosterone undecanoate, or testosterone 17β-undecanoate, is a synthetic androstane steroid and a derivative of testosterone.[33][34] It is an androgen ester; specifically, it is the C17β undecylate (undecanoate) ester of testosterone.[33][34] A related testosterone ester with a similarly very long duration is testosterone buciclate.[8][9]
History
In the late 1970s, testosterone undecanoate was introduced for oral use in Europe,[17] although intramuscular testosterone undecanoate had already been in use in China for several years.[18] Intramuscular testosterone undecanoate was not introduced in Europe and the United States until much later, in the early to mid 2000s and 2014, respectively.[19][20] Testosterone undecanoate was approved in the United States only in 2014 after three previous rejections due to safety concerns.[35]
Society and culture
Generic names
Testosterone undecanoate is the generic name of the drug and its USAN and BAN.[33][34][36][37] It is also referred to as testosterone undecylate.[33][34][36][37]
Brand names
Testosterone undecanoate is or has been marketed under a variety of brand names, including Andriol, Androxon, Aveed, Cernos Depot, Jatenzo, Kyzatrex,[5] Nebido, Nebido-R, Panteston, Reandron 1000, Restandol, Sustanon 250, Undecanoate 250, and Undestor.[33][34][36][38][37]
Availability
Oral testosterone undecanoate is available in Europe, Mexico, Asia, and in the United States.[39][40]
Intramuscular testosterone undecanoate has approved in over 100 countries worldwide,[39][10] including European Union, Russia and the USA.[10][39][41] Intramuscular testosterone undecanoate is marketed most commonly as Nebido in Europe and as Aveed in the United States while oral testosterone undecanoate is marketed most commonly as Andriol.[10][39][41] However, as of 2023 Nebido is not on the formulary of most US hospitals, due to several incidents of pulmonary embolism that have been reported in the country.
Legal status
Testosterone undecanoate, along with other AAS, is a schedule III controlled substance in the United States under the Controlled Substances Act and a schedule IV controlled substance in Canada under the Controlled Drugs and Substances Act.[42][43]
In March 2019, the US Food and Drug Administration approved testosterone undecanoate (Jatenzo), an oral testosterone capsule to treat men with certain forms of hypogonadism. These men have low testosterone levels due to specific medical conditions, such as genetic disorders like Klinefelter syndrome or tumors that have damaged the pituitary gland.[21] The FDA granted the approval of Jatenzo to Clarus Therapeutics.[21][44]
In March 2022, testosterone undecanoate (Tlando) was approved for medical use in the United States.[4]
In July 2022, Kyzatrex, an oral testosterone undecanoate capsule, was approved for medical use in the United States.[5] The FDA granted the approval of Kyzatrex to Marius Pharmaceuticals.[45]
Research
Non-alcoholic steatohepatitis
In 2013, a phase II clinical trial testing intramuscular testosterone undecanoate for the treatment of non-alcoholic steatohepatitis (NASH) was initiated in the United Kingdom.[46] In the United States in 2018, Lipocine Inc. began investigating the potential of using an oral testosterone undecanoate formulation, known as LPCN-1144, in patients with NASH.[47]
Osteoporosis
In 2013, a study aimed to evaluate the efficacy of testosterone undecanoate therapy on bone mineral density (BMD) and biochemical markers of bone turnover in elderly males with osteoporosis and low serum testosterone levels.
They study found that administering low-dose testosterone undecanoate (TU) at a rate of 20 mg per day to elderly men with low serum testosterone and osteoporosis effectively increases bone mineral density in the lumbar spine and femoral neck, and improves bone turnover, similar to the standard-dose TU (40 mg, per day) treatment. The treatment did not exhibit any adverse side effects on the prostate gland, including prostate-specific antigen. Therefore, low-dose TU appears to be a safe and cost-effective protocol for treating elderly male osteoporosis.[48] However, further clinical trials with larger sample sizes, multiple centers, and long-term follow-ups are required to determine the efficacy and safety of low-dose testosterone undecanoate treatment in elderly male osteoporosis with low serum testosterone.
Health implications
Body composition
In 2020, a study that evaluated the effects of testosterone therapy in men with testosterone deficiency and varying degrees of weight (normal weight, overweight, and obesity) on anthropometric and metabolic parameters found that long-term testosterone undecanoate therapy in hypogonadal men, regardless of their weight at the start of the study, led to improvements in several body composition parameters, including body weight, waist circumference, and body mass index. Additionally, testosterone undecanoate therapy was found to lower fasting blood glucose and HbA1c levels and improve lipid profiles in this population.[49]
Bone density
There have been several studies that evaluate the effect of testosterone therapy on bone density or bone mineral density (BMD). One study concluded that long-term testosterone replacement therapy (TRT) in middle-aged men with late-onset hypogonadism (LOH) and metabolic syndrome (MS) led to a significant increase in both vertebral and femoral bone mineral density (BMD) after 36 months of treatment, as measured by dual-energy x-ray absorptiometry. The TRT treatment was shown to induce a 5% per year increase in BMD without changes in body mass index (BMI). The study suggests that long-term TRT could be beneficial for improving bone health in middle-aged men with LOH and MS, even in the absence of osteoporosis.[50]
References
- ↑ "Testosterone Use During Pregnancy". Drugs.com. 20 August 2019. Archived from the original on 1 February 2014. Retrieved 18 March 2020.
- ↑ "FDA-sourced list of all drugs with black box warnings (Use Download Full Results and View Query links.)". nctr-crs.fda.gov. FDA. Retrieved 22 Oct 2023.
- 1 2 "Jatenzo- testosterone undecanoate capsule, liquid filled". DailyMed. 11 January 2021. Archived from the original on 5 January 2022. Retrieved 27 May 2022.
- 1 2 3 "Tlando- testosterone undecanoate capsule, liquid filled". DailyMed. 28 March 2022. Archived from the original on 3 July 2022. Retrieved 27 May 2022.
- 1 2 3 "Kyzatrex- testosterone undecanoate capsule, liquid filled". DailyMed. 18 October 2022. Retrieved 21 January 2023.
- 1 2 3 4 5 6 7 8 Nieschlag E, Behre HM, Nieschlag S (13 January 2010). Andrology: Male Reproductive Health and Dysfunction. Springer Science & Business Media. pp. 441–446. ISBN 978-3-540-78355-8.
- 1 2 3 4 5 Behre HM, Abshagen K, Oettel M, Hübler D, Nieschlag E (May 1999). "Intramuscular injection of testosterone undecanoate for the treatment of male hypogonadism: phase I studies". European Journal of Endocrinology. 140 (5): 414–419. CiteSeerX 10.1.1.503.1752. doi:10.1530/eje.0.1400414. PMID 10229906. S2CID 22597244.
- 1 2 3 4 Nieschlag E, Behre HM, Nieschlag S (26 July 2012). Testosterone: Action, Deficiency, Substitution. Cambridge University Press. pp. 313–315, 321–322. ISBN 978-1-107-01290-5. Archived from the original on 7 April 2022. Retrieved 3 January 2018.
- 1 2 3 Becker KL (2001). Principles and Practice of Endocrinology and Metabolism. Lippincott Williams & Wilkins. pp. 1185–. ISBN 978-0-7817-1750-2. Archived from the original on 2020-05-17. Retrieved 2018-01-03.
- 1 2 3 4 5 6 7 8 9 10 11 12 13 Llewellyn W (2011). Anabolics. Molecular Nutrition LLC. pp. 180–182, 331–334. ISBN 978-0-9828280-1-4. Archived from the original on 2022-07-03. Retrieved 2018-01-03.
- ↑ Irwig MS (April 2017). "Testosterone therapy for transgender men". The Lancet. Diabetes & Endocrinology. 5 (4): 301–311. doi:10.1016/S2213-8587(16)00036-X. PMID 27084565.
- ↑ Jacobeit JW, Gooren LJ, Schulte HM (September 2007). "Long-acting intramuscular testosterone undecanoate for treatment of female-to-male transgender individuals". The Journal of Sexual Medicine. 4 (5): 1479–1484. doi:10.1111/j.1743-6109.2007.00556.x. PMID 17635694.
- ↑ Jacobeit JW, Gooren LJ, Schulte HM (November 2009). "Safety aspects of 36 months of administration of long-acting intramuscular testosterone undecanoate for treatment of female-to-male transgender individuals". European Journal of Endocrinology. 161 (5): 795–798. doi:10.1530/EJE-09-0412. PMID 19749027.
- 1 2 3 4 Bertelloni S, Hiort O (28 September 2010). New Concepts for Human Disorders of Sexual Development. S. Karger AG. pp. 256–. ISBN 978-3-8055-9569-8. Archived from the original on 8 November 2020. Retrieved 3 November 2016.
- 1 2 Kicman AT (June 2008). "Pharmacology of anabolic steroids". British Journal of Pharmacology. 154 (3): 502–521. doi:10.1038/bjp.2008.165. PMC 2439524. PMID 18500378.
- ↑ Santoro N, Braunstein GD, Butts CL, Martin KA, McDermott M, Pinkerton JV (April 2016). "Compounded Bioidentical Hormones in Endocrinology Practice: An Endocrine Society Scientific Statement". The Journal of Clinical Endocrinology and Metabolism. 101 (4): 1318–1343. doi:10.1210/jc.2016-1271. PMID 27032319.
- 1 2 Hoberman J (21 February 2005). Testosterone Dreams: Rejuvenation, Aphrodisia, Doping. University of California Press. pp. 134–. ISBN 978-0-520-93978-3.
- 1 2 Mundy AR, Fitzpatrick J, Neal DE, George NJ (26 July 2010). The Scientific Basis of Urology. CRC Press. pp. 294–. ISBN 978-1-84184-749-8. Archived from the original on 14 April 2019. Retrieved 27 October 2017.
- 1 2 Melmed S, Polonsky KS, Larsen PR, Kronenberg MH (11 November 2015). Williams Textbook of Endocrinology. Elsevier Health Sciences. pp. 709, 711, 765. ISBN 978-0-323-34157-8. Archived from the original on 14 April 2019. Retrieved 27 October 2017.
- 1 2 "Testosterone Undecanoate-Schering AG". Drugs in R&D. 5 (6): 368–369. 2004. doi:10.2165/00126839-200405060-00012. PMID 15563244. S2CID 43349541.
- 1 2 3 Meyer L (27 March 2019). "FDA approves new oral testosterone capsule for treatment of men with certain forms of hypogonadism" (Press release). U.S. Food and Drug Administration (FDA). Archived from the original on 7 September 2020. Retrieved 27 August 2020.
- ↑ Köhn FM, Schill WB (November 2003). "A new oral testosterone undecanoate formulation". World Journal of Urology. 21 (5): 311–315. doi:10.1007/s00345-003-0372-x. PMID 14579074. S2CID 23627346.
- ↑ Ahmad, Syed W et al. “Is Oral Testosterone the New Frontier of Testosterone Replacement Therapy?.” Cureus vol. 14,8 e27796. 8 Aug. 2022, doi:10.7759/cureus.27796
- ↑ "Testosterone undecanoate depot injection". AdisInsight. Springer Nature Switzerland AG. Archived from the original on 2017-12-11. Retrieved 2017-12-11.
- ↑ "Testosterone undecanoate - Organon". AdisInsight. Springer Nature Switzerland AG. Archived from the original on 2017-12-11. Retrieved 2017-12-11.
- ↑ "Aveed- testosterone undecanoate injection". DailyMed. 23 August 2021. Archived from the original on 5 January 2022. Retrieved 27 May 2022.
- ↑ https://www.bayer.com/sites/default/files/NEBIDO_EN_PIL.pdf
- ↑ Fourcroy JL (27 October 2008). Pharmacology, Doping and Sports: A Scientific Guide for Athletes, Coaches, Physicians, Scientists and Administrators. Routledge. p. 25. ISBN 978-1-134-08880-5. Archived from the original on 3 July 2022. Retrieved 3 November 2016.
- ↑ Ong GS, Somerville CP, Jones TW, Walsh JP (2012). "Anaphylaxis triggered by benzyl benzoate in a preparation of depot testosterone undecanoate". Case Reports in Medicine. 2012: 384054. doi:10.1155/2012/384054. PMC 3261473. PMID 22272209. 384054.
- ↑ "Nebido Monograph – Information for Health Care Professionals". Bayer. 2016. Archived from the original on 19 October 2016. Retrieved 19 October 2016.
- 1 2 Payne AH, Hardy MP (28 October 2007). The Leydig Cell in Health and Disease. Springer Science & Business Media. pp. 423–. ISBN 978-1-59745-453-7. Archived from the original on 10 November 2020. Retrieved 6 October 2016.
- ↑ Yeung SC, Escalante CP, Gagel RF (2009). Medical Care of Cancer Patients. PMPH-USA. pp. 247–. ISBN 978-1-60795-008-0. Archived from the original on 2019-04-14. Retrieved 2017-10-30.
- 1 2 3 4 5 Elks J (14 November 2014). The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies. Springer. pp. 641–642. ISBN 978-1-4757-2085-3. Archived from the original on 22 October 2020. Retrieved 6 October 2016.
- 1 2 3 4 5 Index Nominum 2000: International Drug Directory. Taylor & Francis. January 2000. pp. 1002–1004. ISBN 978-3-88763-075-1. Archived from the original on 2020-01-25. Retrieved 2017-12-13.
- ↑ Tucker ME (March 7, 2014). "FDA Approves Aveed Testosterone Jab, with Restrictions". Medscape. Archived from the original on February 1, 2017. Retrieved December 13, 2016.
- 1 2 3 Morton IK, Hall JM (6 December 2012). Concise Dictionary of Pharmacological Agents: Properties and Synonyms. Springer Science & Business Media. ISBN 978-94-011-4439-1. Archived from the original on 19 August 2020. Retrieved 6 October 2016.
- 1 2 3 "Testosterone". Drugs.com. Archived from the original on 2016-11-13. Retrieved 2017-10-27.
- ↑ "Testosterone undecanoate profile and most popular brands in USA". Downsizefitness.com. Archived from the original on 2022-07-03. Retrieved 2020-09-06.
- 1 2 3 4 Nieschlag E, Nieschlag S (2017). "The History of Testosterone and the Testes: From Antiquity to Modern Times". Testosterone. Springer. pp. 1–19. doi:10.1007/978-3-319-46086-4_1. ISBN 978-3-319-46084-0.
- ↑ Arnold AP, Pfaff DW, Etgen AM, Fahrbach SE, Rubin RT (10 June 2002). Hormones, Brain and Behavior, Five-Volume Set. Academic Press. pp. 20–. ISBN 978-0-12-532104-4. Archived from the original on 22 November 2020. Retrieved 27 October 2017.
- 1 2 "Drug Product Database - Health Canada". Health Canada. March 18, 2010. Archived from the original on November 19, 2016. Retrieved November 13, 2016.
- ↑ Karch SB (21 December 2006). Drug Abuse Handbook, Second Edition. CRC Press. pp. 30–. ISBN 978-1-4200-0346-8. Archived from the original on 14 April 2019. Retrieved 11 November 2017.
- ↑ Lilley LL, Snyder JS, Collins SR (5 August 2016). Pharmacology for Canadian Health Care Practice. Elsevier Health Sciences. p. 50. ISBN 978-1-77172-066-3. Archived from the original on 14 April 2019. Retrieved 11 November 2017.
- ↑ "Clarus Therapeutics Receives U.S. FDA Approval of Jatenzo (Testosterone Undecanoate Capsules for Oral Use) (CIII) for Testosterone Replacement Therapy in Certain Adult Men". Clarustherapeutics.com. Archived from the original on 19 February 2020. Retrieved 14 March 2022.
- ↑ "Kyzatrex (testosterone undecanoate) FDA Approval History". Drugs.com. Retrieved 2023-04-06.
- ↑ "Pilot Open Study of Testosterone Replacement in Non-alcoholic Steatohepatitis". Clinicaltrials.gov. 25 April 2017. Archived from the original on 9 April 2021. Retrieved 14 March 2022.
- ↑ "Lipocine Inc. - Clinical Trials". Lipocine.com. Archived from the original on 24 May 2019. Retrieved 14 March 2022.
- ↑ Wang YJ, Zhan JK, Huang W, Wang Y, Liu Y, Wang S, et al. (2013). "Effects of low-dose testosterone undecanoate treatment on bone mineral density and bone turnover markers in elderly male osteoporosis with low serum testosterone". International Journal of Endocrinology. 2013: 570413. doi:10.1155/2013/570413. PMC 3603196. PMID 23533404.
- ↑ Saad F, Doros G, Haider KS, Haider A (June 2020). "Differential effects of 11 years of long-term injectable testosterone undecanoate therapy on anthropometric and metabolic parameters in hypogonadal men with normal weight, overweight and obesity in comparison with untreated controls: real-world data from a controlled registry study". International Journal of Obesity. 44 (6): 1264–1278. doi:10.1038/s41366-019-0517-7. PMC 7260126. PMID 32060355. S2CID 211102413.
- ↑ Aversa A, Bruzziches R, Francomano D, Greco EA, Fornari R, Di Luigi L, et al. (June 2012). "Effects of long-acting testosterone undecanoate on bone mineral density in middle-aged men with late-onset hypogonadism and metabolic syndrome: results from a 36 months controlled study". The Aging Male. 15 (2): 96–102. doi:10.3109/13685538.2011.631230. PMID 22439807. S2CID 6594250.