 | |
 | |
| Names | |
|---|---|
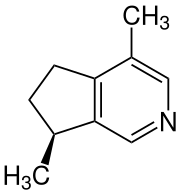
| Preferred IUPAC name
(7S)-4,7-Dimethyl-6,7-dihydro-5H-cyclopenta[c]pyridine | |
| Identifiers | |
3D model (JSmol) |
|
| 81308 | |
| ChEBI | |
| ChemSpider | |
| KEGG | |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C10H13N | |
| Molar mass | 147.221 g·mol−1 |
| Melting point | < 25 °C (77 °F; 298 K) |
| Boiling point | 100 to 103 °C (212 to 217 °F; 373 to 376 K) at 9 mmHg[1] |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
Actinidine is an iridoid produced in nature by a wide variety of plants and animals. It was the first cyclopentanoid monoterpene alkaloid to be discovered.[2] It is one of several compounds that may be extracted from the valerian (Valeriana officinalis) root[3] and silver vine (Actinidia polygama), as well as several types of insects in the larval and imaginal stages.[4] Actinidine is a cat attractant, with effects like those of nepetalactone, the active compound found in catnip.[5]
Certain species of stick insects, including Megacrania batesii and Megacrania tsudai, possess a chemical defense mechanism which involves the secretion of an actinidine-containing substance from the prothoracic glands, when threatened by a predator.[6]
Biosynthesis
A potential biosynthesis of actinidine from L-citronellal is shown below.[7]

References
- ↑ Sakan, Takeo (1959). "On the Structure of Actinidine and Matatabilactone, the Effective Components of Actinidia polygama". Bulletin of the Chemical Society of Japan. 32 (3): 315–316. doi:10.1246/bcsj.32.315.
- ↑ Tsutsui, Minoru; Tsutsui, Ethel Ashworth (1959). "Diterpenoids". Chemical Reviews. 59 (6): 1031–1075. doi:10.1021/cr50030a003.
- ↑ Janot MM, Guilhem J, Contz O, Venera G, Cionga E (1979). "Contribution to the study of valerian alcaloids (Valeriana officinalis L.): actinidine and naphthyridylmethylketone, a new alkaloid". Ann. Pharm. Fr. (in French). 37 (9–10): 413–20. PMID 547813.
- ↑ Weibel DB, Oldham NJ, Feld B, Glombitza G, Dettner K, Boland W (2001). "Iridoid biosynthesis in staphylinid rove beetles (Coleoptera: Staphylinidae, Philonthinae)". Insect Biochemistry and Molecular Biology. 31 (6–7): 583–591. doi:10.1016/s0965-1748(00)00163-6. PMID 11267897.
- ↑ Lichman, Benjamin R.; Godden, Grant T.; Hamilton, John P.; Palmer, Lira; Kamileen, Mohamed O.; Zhao, Dongyan; Vaillancourt, Brieanne; Wood, Joshua C.; Sun, Miao; Kinser, Taliesin J.; Henry, Laura K. (2020-05-01). "The evolutionary origins of the cat attractant nepetalactone in catnip". Science Advances. 6 (20): eaba0721. Bibcode:2020SciA....6..721L. doi:10.1126/sciadv.aba0721. ISSN 2375-2548. PMC 7220310. PMID 32426505.
- ↑ Wu, I‐Hsin; Liu, Hsui‐Huei; Chen, Yu‐Yen; Tsai, Cheng‐Lung; Yu, Yi‐Ching; Hsiao, Chung‐Yi; Yeh, Wen‐Bin (2020). "Life cycles, phenology and genetic structure of endangered Megacrania tsudai Shiraki (Phasmatodea: Phasmatidae): Male individuals from a geographic parthenogenesis species". Entomological Science. 23 (2): 183–192. doi:10.1111/ens.12410. S2CID 216322536.
- ↑ Funayama, Shinji; Cordell, Geoffrey A., eds. (2015). "14: Alkaloids Derived from Terpenoids". Alkaloids. Academic Press. pp. 233–255.