 | |
| Names | |
|---|---|
| Preferred IUPAC name
1,2,4,5-Tetrabromobenzene | |
| Identifiers | |
3D model (JSmol) |
|
| ECHA InfoCard | 100.010.231 |
| EC Number |
|
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C6H2Br4 | |
| Appearance | white solid |
| Density | 2.518 g/cm3 |
| Melting point | 180–182 °C (356–360 °F; 453–455 K) |
| Hazards | |
| GHS labelling: | |
 | |
| Warning | |
| H315, H319, H335, H413 | |
| P261, P264, P271, P273, P280, P302+P352, P304+P340, P305+P351+P338, P312, P321, P332+P313, P337+P313, P362, P403+P233, P405, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
1,2,4,5-Tetrabromobenzene is a bromobenzene compound with the formula C6H2Br4. It is one of three isomers of tetrabromobenzene. The compound is a white solid. 1,2,4,5-Tetrabromobenzene is an important metabolite of the flame retardant hexabromobenzene.[1]
Preparation
The synthesis of 1,2,4,5-tetrabromobenzene has already been reported in 1865 from benzene and excess bromine in a sealed tube at 150 °C.[2] However, the clearly reduced melting point of about 160 °C indicates impurities in the final product. In his 1885 dissertation, Adolf Scheufelen published the synthesis of a purer sample using iron(III) chloride FeCl3 as a catalyst, isolated as "pretty needles" ("schönen Nadeln").[3]
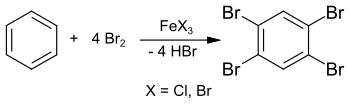
The synthesis can also be carried out in solution in chloroform or tetrachloromethane and yields 1,2,4,5-tetrabromobenzene in 89% yield.[4] This reaction can also be carried out in a laboratory experiment with excess bromine and iron nails (as starting material for iron (III) bromide FeBr3).[5] The intermediate stage is 1,4-dibromobenzene, which reacts further with excess bromine to give 1,2,4,5-tetrabromobenzene.
Reactions
Building block for liquid crystals and fluorescent dyes
Owing to its symmetrical structure and reactivity, 1,2,4,5-tetrabromobenzene is a precursor to nematic liquid crystals[6] with crossed mesogens and for columnar (discotic) liquid crystals[7][8] with an extensive planar, "board-like" tetrabenzoanthracene core.

In a one-pot reaction, 1,2,4,5-tetrabromobenzene reacts with 4-hydroxybenzaldehyde, the alkylating agent 1-bromopentane, the Wittig reagent methyltriphenylphosphonium iodide, the base potassium carbonate, the phase transfer catalyst tetrabutylammonium bromide, the Heck reagent palladium(II)acetate and the Heck co-catalyst 1,3-bis(diphenylphosphino)propane (dppp) in dimethylacetamide obtaining directly a symmetrical tetraalkoxylstilbene as E-isomer in 17% yield.[9]
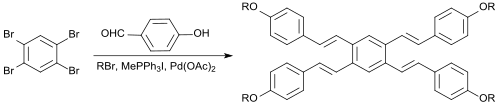
Due to their pronounced π-conjugation such compounds could be potentially applied as optical brighteners, OLED materials or liquid crystals.
N-alkyl-tetraaminobenzenes are available from 1,2,4,5-tetrabromobenzene in high yields, which can be cyclized with triethyl orthoformate and acids to benzobis(imidazolium) salts (BBI salts) and oxidized with oxygen to form 1,4-benzoquinone diimines.[10]
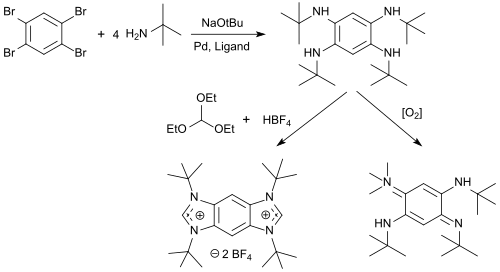
BBI salts are versatile fluorescent dyes with emission wavelengths λem between 329 and 561 nm, pronounced solvatochromism and strong solvent-dependent Stokes shift, which can be used as protein tag for fluorescent labeling of proteins.[11]
Starting material for arynes
From 1,2,4,5-tetrabromobenzene, a 1,4-monoarine can be prepared in-situ with one equivalent of n-butyllithium by bromine abstraction, which reacts immediately with furan to form 6,7-dibromo-1,4-epoxy-1,4-dihydronaphthalene (6,7-dibromonaphthalene-1,4-endoxide) in 70% yield.[12]

When 2,5-dialkylfurans (e.g. 2,5- (di-n-octyl)furan) are used, the dibrominated monoendoxide is formed in 64% yield, from which dibromo-5,8-di-n-octylnaphthalene is formed with zink powder/titanium tetrachloride in 88% yield.[13]
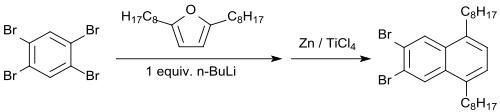
Upon treatment with titanium tetrachloride and zinc dust, the endoxide is deoxygenated yielding 2,3-dibromnaphthalene.[14]
The endoxide reacts with 3-sulfolene in a Diels-Alder reaction upon elimination of sulfur dioxide. The resulting tricyclic adduct converts to 2,3-dibromoanthracene in good yield.[15]
If the dibromene oxide is allowed to react further with furan, in the presence of n-butyllithium[12] or potassium amide[16] or via an intermediate 1,4-aryne the tricyclic 1,4-adduct 1,4:5.8-diepoxy-1,4,5,8-tetrahydroanthracene[17] is formed in 71% yield as a syn-anti-mixture. With sodium amide in ethylene glycol dimethyl ether (DME), however, the dibromene oxide behaves as a 1,3-aryne equivalent and forms with furan a phenanthrene-like tricyclic 1,3-adduct, which can react with furan and sodium amide to a triphenylene derivative (1,3,5-tris-arene).[16]
[2+4] cycloadditions with 1,2,4,5-tetrabromobenzene sometimes proceed in very high yields, such as the reaction of a dihalogen-substituted 1,3-diphenyl-isobenzofuran to a tetrahalogenated anthracene derivative (98%), which is converted successively further with 1,3-diphenyl isobenzofuran in 65% yield to a pentacene derivative and furan to a hexacene derivative (67%).[18]
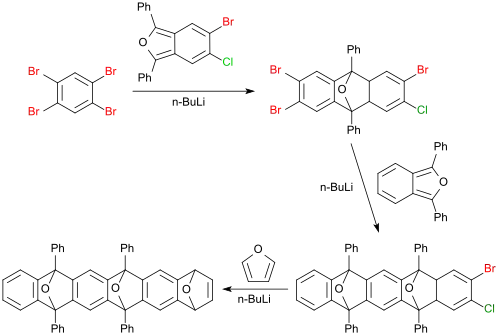
The crosslinking of benzimidazole-modified polymers provides materials with a high absorption capacity for carbon dioxide, which could be suitable for CO2 separation from gas mixtures.[19]
It is the starting material for mono- and bis-aryines.[12]
Safety
1,2,4,5-Tetrabromobenzene is a liver toxic degradation product of the flame retardant hexabromobenzene and was already in 1987 detected in Japan in mother's milk samples.[20]
References
- ↑ E. Bruchajzer; B. Frydrych; J.A. Szymanska (2004), "Effect of repeated administration of hexabromobenzene and 1,2,4,5-tetrabromobenzene on the levels of selected cytochromes in rat liver", Int. J. Occup. Med. Environ. Health, vol. 17, no. 3, pp. 347–353, doi:10.1016/S0040-4020(01)87581-5, PMID 15683155
- ↑ A. Riche, P. Bérard (1865), "Ueber die bromhaltigen Derivate des Benzols und seiner Homologen", Liebigs Ann. Chem. (in German), vol. 133, no. 1, pp. 51–54, doi:10.1002/jlac.18651330106
- ↑ A. Scheufelen (1885), "Ueber Eisenverbindungen als Bromüberträger", Liebigs Ann. Chem. (in German), vol. 231, no. 2, pp. 152–195, doi:10.1002/jlac.18852310204
- ↑ US 0
- ↑ B. Cox; D.G. Kubler; C.A. Wilson (1977), "Experiments with electrophilic aromatic substitution reactions", J. Chem. Educ., vol. 54, no. 6, p. 379, doi:10.1021/ed054p379
- ↑ H.-H. Chen; et al. (2012), "Enantiotropic nematics from cross-like 1,2,4,5-tetrakis(4'-alkyl-4-ethynylbiphenyl) benzenes and their biaxiality studies", Chem. Eur. J., vol. 18, no. 31, pp. 9543–9551, doi:10.1002/chem.201103453, PMID 22745006
- ↑ S. Kumar (2011), Chemistry of discotic liquid crystals: from monomers to polymers, Boca Raton, FL, U.S.A.: CRC Press, p. 200, ISBN 978-1-4398-1145-0
- ↑ M.C. Artal; K.J. Toyne; J.W. Goodby; J. Barbera; D.J. Photinos (2011), "Synthesis and mesogenic properties of novel board-like liquid crystals", J. Mater. Chem., vol. 11, no. 11, pp. 2801–2807, doi:10.1039/B105351P
- ↑ K.N. Patel; B.V. Kamath; A.V. Bedekar (2013), "Synthesis of alkyloxy stilbenes by one-pot O-alkylation-Wittig and O-alkylation-Wittig-Heck reaction sequence", Tetrahedron Lett., vol. 54, no. 1, pp. 80–84, doi:10.1016/j.tetlet.2012.10.102
- ↑ D.M. Khramov; A.J. Boydston; C.W. Bielawski (2006), "Highly efficient synthesis and solid-state characterization of 1,2,4,5-tetrakis(alkyl- and arylamino)benzenes and cyclization to their respective benzobis(imidazolium) salts", Org. Lett., vol. 8, no. 9, pp. 1831–1834, doi:10.1021/ol060349c, PMID 16623562
- ↑ A.J. Boydston (2008), "Modular fluorescent benzobis(imidazolium)saltes: Syntheses, photophysical analyses, and applications", J. Am. Chem. Soc., vol. 130, no. 10, pp. 3143–3156, doi:10.1021/ja7102247, PMID 18271588
- 1 2 3 K. Shahlai; S. O. Acquaah; H. Hart (1998). "USE OF 1,2,4,5-Tetrabromobenzene as a 1,4-Benzadiyne Equivalent: Anti- and Syn-1,4,5,8-Tetrahydroanthracene 1,4:5,8-Diepoxide". Organic Syntheses. 75: 201. doi:10.15227/orgsyn.075.0201.
- ↑ Z. Chen; P. Müller; T.M. Swager (2006), "Syntheses of soluble, π-stacking tetracene derivatives", Org. Lett., vol. 8, no. 2, pp. 273–276, doi:10.1021/ol0526468, PMID 16408893
- ↑ H. Hart; C.-Y. Lai; G.C. Nwokogu; S. Shamouilian (1987), "Tetrahalobenzenes as diaryne equivalents in polycyclic arene synthesis", Tetrahedron, vol. 43, no. 22, pp. 5203–5224, doi:10.1016/S0040-4020(01)87696-1
- ↑ C.-T. Lin; T.-C. Chou (1988), "Synthesis of 2,3-dibromoanthracene", Synthesis, vol. 1988, no. 8, pp. 628–630, doi:10.1055/s-1988-27659, S2CID 93109532
- 1 2 F. Raymo; F.H. Kohnke; F. Cardullo (1992), "The regioselective generation of arynes from polyhalogenobenzenes. An improved synthesis of syn- and anti-1,4,5,8,9,12-hexahydro-1,4:5,8:9,12-triepoxytriphenylene", Tetrahedron (in German), vol. 48, no. 33, pp. 6827–6838, doi:10.1016/S0040-4020(01)89874-4
- ↑ H. Hart; N. Raju; M.A. Meador; D.L. Ward (1983), "Synthesis of heptiptycenes with face-to-face arene rings via a 2,3:6,7-anthradiyne equivalent", J. Org. Chem., vol. 48, no. 23, pp. 4357–4360, doi:10.1021/jo00171a039
- ↑ S. Eda, T. Hamura (2015), "Selective Halogen-Lithium Exchange of 1,2-Dihaloarenes for Successive [2+4] Cycloadditions of Arynes and Isobenzofurans", Molecules, vol. 20, no. 10, pp. 19449–19462, doi:10.3390/molecules201019449, PMC 6331892, PMID 26512641
- ↑ S. Altarawneh; S. Behera; P. Jena; H.M. El-Kaderi (2014), "New insights into carbon dioxide interactions with benzimidazole-linked polymers", Chem. Commun., vol. 50, no. 27, pp. 3571–3574, doi:10.1039/C3CC45901B, PMID 24567950
- ↑ T. Miyazaki; T. Yamagishi; M. Matsumoto (1987), "Determination and residual levels of 1,2,4,5-tetrabromobenzene and Mirex in human milk samples", Food Hygiene and Safety Science, vol. 28, no. 2, pp. 125–129, doi:10.3358/shokueishi.28.125