The Zn/ZnO cycle
For chemical reactions, the zinc–zinc oxide cycle or Zn–ZnO cycle is a two step thermochemical cycle based on zinc and zinc oxide[1] for hydrogen production[2] with a typical efficiency around 40%.[3]
Process description
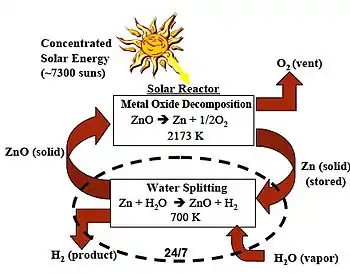
The thermochemical two-step water splitting process uses redox systems:[4]
- Dissociation: ZnO → Zn + 1/2 O2
- Hydrolysis: Zn + H2O → ZnO + H2
For the first endothermic step concentrating solar power is used in which zinc oxide is thermally dissociated at 1,900 °C (3,450 °F) into zinc and oxygen. In the second non-solar exothermic step zinc reacts at 427 °C (801 °F) with water and produces hydrogen and zinc oxide. The temperature level is realized by using a solar power tower and a set of heliostats to collect the solar thermal energy.
See also
References
- ↑ Solar Hydrogen Production from a ZnO/Zn Thermo-chemical Cycle Archived July 24, 2008, at the Wayback Machine
- ↑ Project PD10
- ↑ Novel Method for solar hydrogen generation Archived February 5, 2009, at the Wayback Machine
- ↑ Solar thermal ZnO-decomposition
External links
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.