 | |
| Names | |
|---|---|
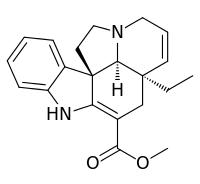
| IUPAC name
Methyl 2,3,6,7-tetradehydro-5α,12β,19α-aspidospermidine-3-carboxylate | |
| Systematic IUPAC name
Methyl (3aR,3a1S,10bR)-3a-ethyl-3a,3a1,4,6,11,12-hexahydro-1H-indolizino[8,1-cd]carbazole-5-carboxylate | |
| Identifiers | |
| |
3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.022.378 |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C21H24N2O2 | |
| Molar mass | 336.435 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
Tabersonine is a terpene indole alkaloid found in the medicinal plant Catharanthus roseus and also in the genus Voacanga[1] (both taxa belonging to the alkaloid-rich family Apocynaceae). Tabersonine is hydroxylated at the 16 position by the enzyme tabersonine 16-hydroxylase (T16H) to form 16-hydroxytabersonine.[2] The enzyme leading to its formation is currently unknown. Tabersonine is the first intermediate leading to the formation of vindoline one of the two precursors required for vinblastine biosynthesis.
See also
References
- ↑ Voacanga, (Apocynaceae), a review of its taxonomy, phytochemistry, ethnobotany and pharmacology. Leeuwenberg et al Agric. Univ. Wagenigen papers #85-3, 1985. Page 8
- ↑ St-Pierre and De Luca (1995) A Cytochrome P-450 Monooxygenase Catalyzes the First Step in the Conversion of Tabersonine to Vindoline in Catharanthus roseus. Plant Physiology. 109(1). 131-139
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.