| |||
| Names | |||
|---|---|---|---|
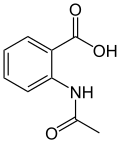
| Preferred IUPAC name
2-Acetamidobenzoic acid | |||
| Other names
2-Acetamidobenzoic acid; 2-Carboxyacetanilide; o-Acetoaminobenozic acid; Acetylanthranilic acid; 2-(Acetylamino)benzoic acid | |||
| Identifiers | |||
3D model (JSmol) |
|||
| ChEBI | |||
| ChemSpider | |||
| ECHA InfoCard | 100.001.741 | ||
PubChem CID |
|||
| UNII | |||
CompTox Dashboard (EPA) |
|||
| |||
| |||
| Properties | |||
| C9H9NO3 | |||
| Molar mass | 179.175 g·mol−1 | ||
| Appearance | Slightly beige solid | ||
| Density | 1.36 g/mL | ||
| Melting point | 184 to 186 °C (363 to 367 °F; 457 to 459 K) | ||
| Boiling point | 399 °C (750 °F; 672 K) | ||
| Hazards | |||
| GHS labelling: | |||
 | |||
| Lethal dose or concentration (LD, LC): | |||
LD50 (median dose) |
Oral, mouse = 1114 mg/kg | ||
| Safety data sheet (SDS) | - | ||
| Legal status | |||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |||
N-Acetylanthranilic acid is an organic compound with the molecular formula C9H9NO3. It is an intermediate product in catabolism of quinaldine in Arthrobacter sp., and is further metabolized to anthranilic acid.[2][3]
N-Acetylanthranilic acid can be synthesized from 2-bromoacetanilide via palladium-catalyzed carbonylation in tri-n-butylamine-water at 110–130 °C, under 3 atm of carbon monoxide.[4] In the laboratory, it can be easily synthesized from anthranilic acid and acetic anhydride.
N-Acetylanthranilic acid exhibits triboluminescence when crushed.[5] The fractured crystals have large electrical potentials between areas of high and low charge. When the electrons suddenly migrate to neutralize these potentials, flashes of deep blue light are created.
In the United States, it is a Drug Enforcement Administration-controlled List I chemical,[6] because it has been used in the synthesis of methaqualone.
See also
References
- ↑ Anvisa (2023-03-31). "RDC Nº 784 - Listas de Substâncias Entorpecentes, Psicotrópicas, Precursoras e Outras sob Controle Especial" [Collegiate Board Resolution No. 784 - Lists of Narcotic, Psychotropic, Precursor, and Other Substances under Special Control] (in Brazilian Portuguese). Diário Oficial da União (published 2023-04-04). Archived from the original on 2023-08-03. Retrieved 2023-08-15.
- ↑ Hund HK, de Beyer A, Lingens F (1990). "Microbial metabolism of quinoline and related compounds. VI. Degradation of quinaldine by Arthrobacter sp". Biol Chem Hoppe-Seyler. 371 (10): 1005–1008. doi:10.1515/bchm3.1990.371.2.1005. PMID 2076195.
- ↑ Overhage J, et al. (2005). "Identification of large linear plasmids in Arthrobacter spp. encoding the degradation of quinaldine to anthranilate". Microbiology. 151 (2): 491–500. doi:10.1099/mic.0.27521-0. PMID 15699198.
- ↑ Donald Valentine; Jefferson W. Tilley; Ronald A. LeMahieu (1981). "Practical, catalytic synthesis of anthranilic acids". Journal of Organic Chemistry. 46 (22): 4614–4617. doi:10.1021/jo00335a075.
- ↑ Erikson J (Oct 1972). "N-acetylanthranilic acid. A highly triboluminescent material". J Chem Educ. 49 (10): 688. doi:10.1021/ed049p688.
- ↑ "PART 1310 - Section 1310.02 Substances covered". www.deadiversion.usdoj.gov.