 | |
| Clinical data | |
|---|---|
| Trade names | Solu-Medrol, Solu-Medrone, Urbason, others |
| Other names | Methylprednisolone hemisuccinate; 6α-Methylprednisolone 21-hemisuccinate |
| AHFS/Drugs.com | Monograph |
| License data | |
| Routes of administration | Intravenous injection |
| Drug class | Corticosteroid; Glucocorticoid |
| ATC code | |
| Legal status | |
| Legal status | |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.017.416 |
| Chemical and physical data | |
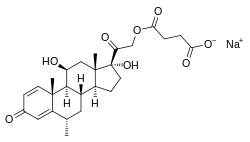
| Formula | C26H34O8 |
| Molar mass | 474.550 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Methylprednisolone succinate, sold under the brand name Solu-Medrol among others, is a synthetic glucocorticoid corticosteroid and a corticosteroid ester—specifically the C21 succinate ester of methylprednisolone—which is used by intravenous administration.[2][3] Methylprednisolone succinate is provided as two different salts when used as a pharmaceutical drug: a sodium salt (methylprednisolone sodium succinate; brand name Solu-Medrol, others) and a hydrogen salt (methylprednisolone hemisuccinate or methylprednisolone hydrogen succinate; brand name Urbason).[2][3]
Methylprednisolone succinate was approved for medical use in the United States in 1959.[1]
References
- 1 2 "Solu-Medrol- methylprednisolone sodium succinate injection, powder, for solution". DailyMed. 17 December 2021. Retrieved 23 December 2023.
- 1 2 Elks J (14 November 2014). The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies. Springer. pp. 811–. ISBN 978-1-4757-2085-3.
- 1 2 Index Nominum 2000: International Drug Directory. Taylor & Francis. 2000. pp. 675–. ISBN 978-3-88763-075-1.
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.