 | |
| Names | |
|---|---|
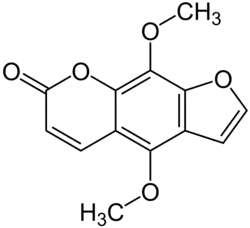
| Preferred IUPAC name
4,9-Dimethoxy-7H-furo[3,2-g][1]benzopyran-7-one | |
| Other names
5,8-Dimethoxypsoralen 5,8-Dimethoxypsoralene | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.166.737 |
| KEGG | |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C13H10O5 | |
| Molar mass | 246.21 g/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
Isopimpinellin is a natural product synthesized by numerous plant species, especially species in the carrot family Apiaceae. The compound can be found in celery, garden angelica, parsnip, fruits and in the rind and pulp of limes.[1] Several studies have looked into the effects of isopimpinellin and other furanocoumarins (such as bergamottin and imperatorin) as anticarcinogens.[1][2] These studies have shown possible inhibition of 7,12-Dimethylbenz(a)anthracene, which are initiators of skin tumors.[1] Evidence has also been reported that links these compounds to the inhibition of breast cancers.[2]
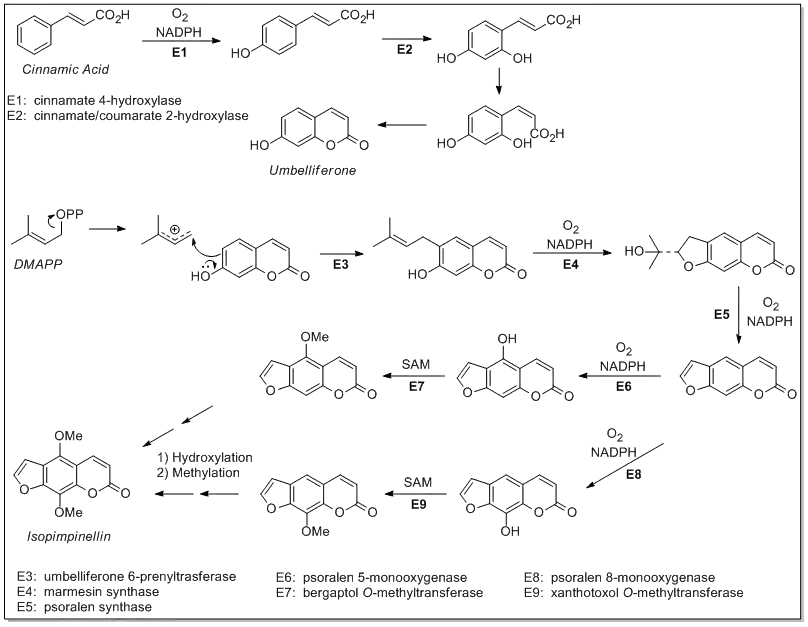
Biosynthesis
Isopimpinellin is a furanocoumarin thought to be synthesized through the mevalonate pathway via addition of dimethylallyl pyrophosphate (DMAPP) to a modified coumarate known as umbelliferone. The biosynthesis is shown below:[3]
References
- 1 2 3 Kleiner, Heather E.; Suryanarayana V.Vulimiri; Matthew F.Starost; Melissa J.Reed; John DiGiovanni (2002). "Oral administration of the citrus coumarin, isopimpinellin, blocks DNA adduct formation and skin tumor initiation by 7,12-dimethylbenz[a]anthracene in SENCAR mice". Carcinogenesis. 23 (10): 1667–1675. doi:10.1093/carcin/23.10.1667. PMID 12376476.
- 1 2 Prince, Misty; Cheryl T.Campbell; Taylor A.Robertson; Amy J.Wells; Heather E.Kleiner (2006). "Naturally occurring coumarins inhibit 7,12-dimethylbenz[a]anthracene DNA adduct formation in mouse mammary gland". Carcinogenesis. 27 (6): 1204–13. doi:10.1093/carcin/bgi303. PMID 16387742.
- ↑ Dewick, Paul M. (2009). Medicinal Natural Products: A Biosynthetic Approach (3rd ed.). UK: John Wiley & Sons Ltd. ISBN 978-0-471-97478-9.