 | |
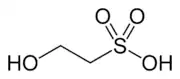
 Isethionic acid | |
| Names | |
|---|---|
| Preferred IUPAC name
2-Hydroxyethane-1-sulfonic acid | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.003.169 |
| KEGG | |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C2H6O4S | |
| Molar mass | 126.13 g/mol |
| Density | 1.63 g/cm3 |
| Acidity (pKa) | 1.39 (predicted) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
Isethionic acid is an organosulfur compound containing an alkylsulfonic acid located beta to a hydroxy group. Its discovery is generally attributed to Heinrich Gustav Magnus, who prepared it by the action of solid sulfur trioxide on ethanol in 1833.[1] It is a white water-soluble solid used in the manufacture of certain surfactants and in the industrial production of taurine. It is most commonly available in the form of its sodium salt (sodium isethionate).
Synthesis
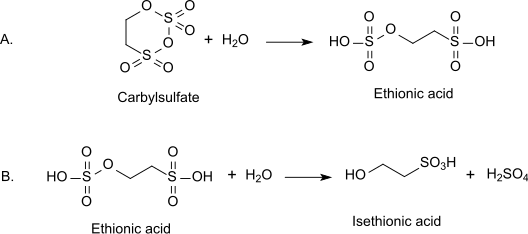
The original synthesis of the compound, involving the reaction of sulfur trioxide with ethanol, has largely been replaced by more advanced methods. An alternative production method involves the hydrolysis of carbyl sulfate, which is derived from the sulfonation of ethylene.
However the most common route is the reaction of ethylene oxide with aqueous sodium bisulfite, which produces the sodium salt (sodium isethionate):
Reactions
Isethionic acid is used as a starting material in the industrial production of taurine.
Dehydration of isethionic acid gives vinylsulfonic acid.[2]
Derivatives
Fatty acid esters of isethionic acid (such as sodium lauroyl isethionate and sodium cocoyl isethionate) are used as biodegradable anionic surfactants.[3] These materials are much milder to skin that other sulfate based surfactants (i.e. sodium lauryl sulfate)[4] making them popular for use in make-up, shampoos and detergent bars including those made by Dove.
Isethionic acid is also used as a counter ion in certain pharmaceutical formulations, including the antimicrobials hexamidine and pentamidine.[5]
Biological importance
Studies made on dog heart slices suggested that heart tissue may be capable of converting taurine to isethionic acid, further experiments demonstrated that this tissue may synthesize taurine from cystine.[6]
See also
References
- ↑ Magnus, G. (1833). "Ueber die Weinschwefelsäure, ihren Einfluss auf die Aetherbildung, und über zwei neue Säuren ähnlicher Zusammensetzung". Annalen der Physik und Chemie. 103 (2): 367–388. Bibcode:1833AnP...103..367M. doi:10.1002/andp.18331030213. ISSN 0003-3804.
- ↑ Kosswig, Kurt (2000). "Sulfonic Acids, Aliphatic". Ullmann's Encyclopedia of Industrial Chemistry. Wiley-VCH. doi:10.1002/14356007.a25_503. ISBN 3-527-30673-0.
- ↑ Petter, P. J. (1984). "Fatty acid sulfoalkyl amides and esters as cosmetic surfactants". International Journal of Cosmetic Science. 6 (5): 249–260. doi:10.1111/j.1467-2494.1984.tb00382.x. ISSN 0142-5463. PMID 19467117. S2CID 41819056.
- ↑ Tupker, R. A.; Bunte, E. E.; Fidler, V.; Wlechers, J. W.; Coenraads, P. J. (1999). "Irritancy ranking of anionic detergents using one-time occlusive, repeated occlusive and repeated open tests". Contact Dermatitis. 40 (6): 316–322. doi:10.1111/j.1600-0536.1999.tb06082.x. ISSN 0105-1873. PMID 10385334. S2CID 418996.
- ↑ Wicho, H, ed. (2009). Austria-Codex Stoffliste (in German) (42 ed.). Vienna: Österreichischer Apothekerverlag.
- ↑ W.O Read and J.D.Welty (1961). "Synthesis of Taurine and Isethionic Acid by Dog Heart Slices". The Journal of Biological Chemistry. 237 (5): 1521–1522. doi:10.1016/S0021-9258(19)83734-1. PMID 14490797.