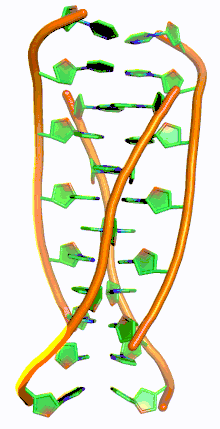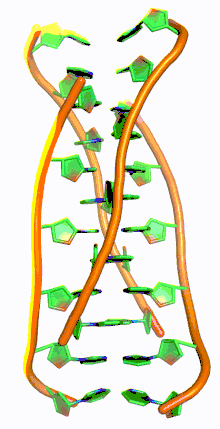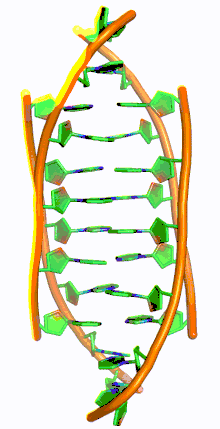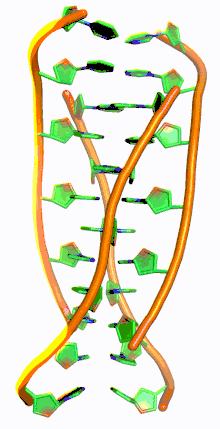
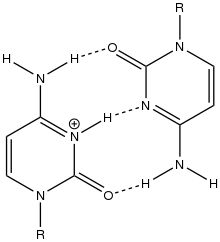
i-motif DNA, short for intercalated-motif DNA, are cytosine-rich four-stranded quadruplex DNA structures, similar to the G-quadruplex structures that are formed in guanine-rich regions of DNA.
History
This structure was first discovered in 1993 by Maurice Guéron at École Polytechnique in Palaiseau, France. It was found when two antiparallel doubled stranded DNA complexes with cytosine-protonated cytosine (C·C*) base pairs became associated with one another. This formed a complex 4- stranded DNA complex.[1] The structure was originally found only in vitro, usually at a slightly acidic pH, but was recently discovered in the nuclei of human cells.[2] A new antibody fragment was created, and was found to have highly specific binding affinity for I-motif complexes, but did not bind to other DNA structures, making it optimal for identifying i-motif structures in cells.[3]
During their media release in April 2018, Dr. Mahdi Zeraati and colleagues mentioned that these complexes are constantly forming and dissociating due to their constantly changing temperatures, which could play a role in its function in regulation of gene expression and cell reproduction. Although the exact function of these structures is unknown, the transient nature of these molecules gives insight regarding the biological function of these molecules. Found primarily in the G1 phase of the cell cycle and in promoter regions, i-motif complexes could potentially affect which gene sequences are read and could play a role in determining which genes are switched on or off.[4] Other experimentation is in progress to determine the role of i-motif DNA in nanotechnology using i-motifs as biosensors and nanomachines,[5] and it has even been seen to play a role in the advancement of cancer therapy.
Structural overview
Similar to G-quadruplex DNA structures with intercalated guanine residues, i-motifs consists of antiparallel tracts of oligodeoxynucleotides strands that contain mostly cytosine residues. The interactions between these molecules occur by the hemi protonation of cytosine residues and non-Watson Crick base pairing, more specifically Hoogsteen base pairing. There are two main intercalated topologies that i-motifs can be classified in: 3'-E, when the outmost C:C+ base pair is at the 3'-end, and 5'-E, where the outermost C:C+ base pair is at the 5'-end. When comparing the two topologies, the 3'-E topology is more stable due to increased sugar-sugar contacts.[9] This occurs due to the difference in Van der Waals energy contribution between the two topologies. The interactions of the sugar-sugar contacts along the narrow grooves allows for optimal backbone twisting, which ultimately contributes to formation of stacking bases and the stability of the molecule.[10] However, the overall stability of i-motif structures is dependent on the number of cytosine residues that are interacting with each other. This means that as more cytosine residues interact through hydrogen bonding, the more stable the molecule will be.[11] Other factors that affect the stability of the molecules include temperature, salt concentration and pH of the environment.[12]
While many i-motif complexes are most stable at a slightly acidic pH (between 4.2 and 5.2),[11] some i-motifs have been found to form at neutral pH, when a free proton is used by the nucleic acids during the folding process. These particular i-motif complexes are found under particular conditions, including low temperature (4 °C), molecular crowding, negative super helicity, and the introduction of silver(I) cations. Maintaining a negative super helicity is crucial for the stabilization of i-motifs at a neutral pH.[13]
i-motif structures have also been found to form under biological conditions. These structures have been discovered in many different locations of the cell including the nuclei,[2] the cytoplasm, and in telomeres and promoter sights.[14] It can also be found in cell processes such as the G1 phase of the cell cycle.
Stability of i-motif DNA
As a nucleic acid structure, i-motif DNA stability is dependent on the nature of the sequence, temperature, and ionic strength. The structural stability of i-motif DNA is mainly reliant on the fact that there is minimal overlap between the six-membered aromatic pyrimidine bases due to the consecutive base pairs' intercalative geometry. Exocyclic carbonyls and amino groups stacked in an antiparallel formation are essential to C:C+ base pairs' stability due to the lack of compensation for the electrostatic repulsion between their charged amino groups.[15] Other factors, including sugar and phosphate backbone interactions, C-tract length, capping and connecting loops interaction, ionic interactions, molecular crowding, and super helicity, all affect the stability of i-motif DNA.[16]
C:C+ base pairs
The C:C+ base pairs contribute most to i-motif stability due to three hydrogen bonds. This stability is exhibited by the base-pairing energy (BPE) of i-motif being 169.7 kJ/mol, which is relatively high compared to neutral C·C and canonical Watson-Crick G·C, which have BPEs of 68.0 kJ/mol and 96.6 kJ/mol, respectively.[17] The most stable central hydrogen bond in the C:C+ base pair (N3··H··N3) has been denoted as having double-well potential due to the proton's capability of oscillating between the two nitrogen base wells[18] with a proton transfer rate found to be 8 × 104 s-1.[19]
The results of two studies by Waller's group and Mir et al. emphasized the importance of electrostatic interactions contributing to the stability of the C:C+ base pair.[20] Waller's group wished to determine the effect of 2 - deoxyriboguanylurea (GuaUre-dR), a chemotherapeutic agent, on i-motif DNA formation in human telomeres. Waller's group found that the addition of GuaUre-dR led to a decrease in pH when compared to i-motifs without it.[21] Mir et al. showed that the addition of pseudoiso-deoxycytidine (psC) increased the stability of head-to-head and head-to-tail dimeric i-motif structures when a neutrally charged psC:C was found at the end of the C:C stack.[22] Both studies ultimately found that the existence of positive charges in the core of these structures contributed most to the stability of the C:C+ base pair.[20]
Alterations to the environment conditions of C:C+ were studied by Watkins et al. to observe changes in overall stability.[23] Chemical modifications to the C:C+ base pair in which halogenated analogs (5-fluoro, 5-bromo, and 5-iodo) took the place of cytosine increased i-motif DNA stability in acidic environments.[24] This study initiated an investigation into the methylation of cytosine and its effect on pH. Methylation of cytosine at position 5 increased pH of mid-transition and Tm of i-motifs. On the other hand, hydroxymethylation leads to a decrease in the pH of mid-transition and Tm.[25]
Phosphate backbone and sugar interactions
The minor groove of i-motif DNA consists of a phosphate backbone in which two negatively charged sides repel each other, requiring balance to stabilize the overall structure.[20] Hydrogen bonding and Van der Waals interactions between sugars of the minor groove from the sequence d(CCCC) of tetrameric i-motif DNA stabilizes the narrow grooves of the i-motif structure. The stability of 3'E and 5'E topologies from the sequence d(CCCC) was observed through molecular dynamic simulations to determine the effect of repulsion between the phosphate backbones.[15] The stability observed in the simulations is derived from the supportive sugar interactions, so much so that the stability of any i-motif is dependent on the balance between sugar interactions and connective loop activity. This is due to the low free energy of the hydrogen bond (CHO) in the i-motif structure with a value of 2.6 kJ/mol.[19]
Modifications to the phosphate backbone have been seen in research in which an alternative to the phosphate backbone was studied. Oligodeoxycytidine phosphorothioates can form intramolecular and intermolecular i-motifs.[26] Mergny and Lacroix determined that the addition of a bulky methyl group had a destabilizing effect on the i-motif formation when they compared phosphorothioate, the natural phosphodiester, methylphosphonate, and peptide linkages and determined that only phosphodiester and phosphorothioate oligodeoxynucleotides were capable of forming stable i-motifs.[27]
Environmental conditions
Investigations into the formation of i-motifs at physiological pH, rather than acidic pH, include simulations of molecular crowding, superhelicitification, and cationic conditions. Through the utilization of polyethylene glycols with a high molecular weight, conditions of congested molecular and nuclear environments were induced.[28] An increase in pKa of cytosine N3[23] showed that these conditions favored quadruplex and i-motif over double[29] and single-stranded DNA when i-motif formation and protonation was Bacollainduced at neutral pH.[30] Negative super helicity assists in the formation of i-motifs under physiological conditions.[31] The formation of both G-quadruplex and i-motifs occurred at neutral pH when G-quadruplex and i-motif forming sequences of the c-MYC oncogene promoter were placed into a supercoiled plasmid, inducing super helicity of both structures. The conditions of super helicity mitigation were inspired by the fact that i-motif destabilizes double-stranded structures.[28][31] This result reflects the transcription process in which supercoiled DNA is unwound into single-stranded structures, which causes negative super helicity.[32] The stability of i-motif DNA can be influenced by increasing ionic concentration.[33] The addition of Na has shown to increase the destabilization of the i-motif structure from the c-jun proto-oncogene at pH 4.8. A decrease in stability of i-motif corresponded with an increase in ionic concentration in a study of i-motif DNA from n-MYC.[34] However, no significant differences in stability occurred with the addition of 5 mM Mg+, Ca+, Zn+, Li+ or K+ cations in the presence of 100 mM NaCl at pH 6.4.[27]
Base modifications
Further investigation is required to determine the absolute effect on i-motif stability when bases are modified, but studies have indicated that there is potential for the modification of bases corresponding to the stability of i-motifs.[35] Two examples include replacing cytosine with 5-methylcytosine[35] and replacing thymine with 5-propynyl uracil,[23] both increasing the stability of the i-motif structure. The modification of bases may be helpful in determining the pH/temperature-dependent folding patterns of i-motifs.[28]
Formation
Intercalated motif (i-motif) DNA is formed in the nuclei of cells via a stack of intercalating hemi-protonated C-neutral C base pairs, which are optimized at a slightly negative pH. In vitro, i-motifs have been characterized with indications that the DNA is derived from telomeres. Using a variety of biophysical techniques, i-motif DNA has been characterized to be derived from centromeres and promoter regions of proto-oncogenes. An analysis of the biophysical results shows the structures' overall stability depends on the number of cytosines in the i-motif core and the length and composition of loops in the formation of both intramolecular and intermolecular structures.[36]
Although it has been largely established that C-rich sequences can form i-motif structures in vitro, there is still significant debate regarding the in vivo existence of four-stranded i-motif DNA structure in the human genome. It has been confirmed that motif DNA in vivo can be formed at physiological pH under certain molecular crowding conditions and negative super helicity induced during transcription.[36] Recent studies have shown that the formation of i-motif DNA by specific genomic sequences can occur at neutral pH. Numerous studies have demonstrated that i-motif DNA affects replication and transcription in DNA processing after its formation.[37]
G-quadruplex formation
I-motif DNA forms from any complementary strand of G-quadruplex forming sequence. G-quadruplexes are helically shaped and found in nucleic acids that are rich in guanine. These secondary structures possess guanine tetrads formed into one of three types of strands: one, two, or four. With prior knowledge of G-quadruplex forming sequences being susceptible to i-motif DNA formation, Waller's group used the algorithm Quadparser to determine the amount of i-motif forming sequences in the human genome.[21] The query consisted of four C-tracts of five cytosines distinguished by the number of nucleotides that could range from 1–19. Across the human genome, 5,125 sequences have potential i-motif formation capabilities with 12.4% (637) of the total resulting sequences found in the promoter regions of genes. Based upon the ontology codes corresponding to the promoter regions, i-motif formation is concentrated sequence-specific DNA binding, DNA templated transcription, skeletal system development and RNA polymerase II positive regulation of transcription.[38]
Interacting agents and ligands
Interacting agents derived from G4 ligands
Tetra-(N-methyl-4-pyridyl)porphyrin (TMPyP4)
The first study to determine a ligand binding to i-motif DNA was by Hurley and colleagues in 2000. They researched the interaction between Tetra-(N-methyl-4-pyridyl)porphyrin (TMPyP4) and tetramolecular i-motif DNA isolated from a human telomeric sequence. The study utilized an electrophoretic mobility shift assay (EMSA) by notably not changing the DNA melting temperature. This ligand interacts with G4 on the i-motif sequence to deregulate c-myc expression and inhibit telomerase.[39][40] Two molecules of TMPyP4 coordinate with i-motif DNA on both the top and bottom of its structure as determined by NMR experiments.[41]
Phenanthroline and acridine derivatives
These cores characterize phenanthroline derivatives due to their G4 binding and telomerase inhibiting activity.[42] This activity leads to an overall increase in the Tm of the i-motif. Phenanthroline derivatives bind to the C:C base pair, leading to a decrease in the binding constant lower than that of a normal G-quadruplex.[43] Acridine derivatives are also G4 ligands and through fluorescence resonance energy transfer (FRET) melting assays, diethylenetriamine (BisA) was determined to increase the melting temperature of both i-motif and G4, while monomeric acridine (MonoA) had no such effect.[44]
Macrocyclic tetraconazoles, L2H2-4OTD
Inspired by telomestatin, a natural potent telomerase inhibitor, macrocyclic poly-oxazoles were synthesized. Macrocyclic poly-oxazole compounds possess the same binding mode as telomestatin when interacting with G4 in a pi-pi stack formation.[43] Smaller macrocycles, penta- (L2H2-5OTD) and tetra-oxazoles (L2H2-4OTD) were developed with amine R-groups to observe stability and binding site locations on i-motif. Reducing the size of the ligands reduced its stabilizing effect on G4-forming sequences. L2H2-4OTD molecules bind cooperatively to Loop 1 and 2 on the telomeres of the i-motif DNA sequence which induces deformities on C3-C15, C2-C14 and C8-C20 base pairs while maintaining the structure of i-motif.[45]
Mitoxantrone, tilorone and tobramycin
Mitoxantrone stabilizes the i-motif and G4 and aids in their formation under neutral conditions with a preference in binding to i-motif over double-stranded DNA. Tilorone and Tobramycin are i-motif binding ligands discovered via thiazole orange fluorescence intensity displacement (FID) assay.[46]
Carboxylic acid-modified single-walled carbon nanotubes (SWCNTs) and graphene quantum dots (GQDs)
SWCNTs stabilize i-motif DNA by attracting water molecules from the structure. GQDs intercalate with DNA to aid in the formation of i-motif DNA by end-stacking loop regions. This process allows GQDs to stabilize i-motifs by minimizing solvent-access.[47]
Ligands used for biological functions
There are several ligands for i-motif that are used for biological functions. These include IMC-48, IMC-76, Nitidine, NSC309874, acridone derivative, and PBP1. IMC-48 stabilizes the bcl-2 structure of i-motif by upregulating bcl-2 gene expression. IMC-76 stabilizes the bcl-2 hairpin structure by downregulating the bcl-2 gene expression. Nitidine destabilizes the hairpin on hybride i-motif/hairpin structure and has no significant interactions with complementary G4. Nitidine downregulates the k-ras gene expression by showing selectivity toward the k-ras structure.[43] NSC309874 stabilizes the PDGFR-b i-motif structure with no significant interaction with complementary G4 to downregulate the PDGFR-b gene expression. Acridone derivative stabilizes the c-myc i-motif structure with no significant interaction with G4 in order to downregulate the c-myc gene expression. PBP1 stabilizes the bcl-2 i-motif structure and promotes its formation in neutral pHs to upregulate the bcl-2 gene expression.[43]
Ligands used as fluorescent probes
The ligands for i-motif used as fluorescent probes include Thiazole orange, 2,2'-diethyl-9-methylselenacarbocyanine bromide (DMSB), crystal violet, berberine neutral red, thioflavin T, and perylene tetracarboxylic acid diimide derivative (PTCDI), originally seen as G4 probes.[43]
Biological function
Large tracts of G/C-rich DNA exist in regulatory regions of genes and in terminal regions of chromosomes and telomeres. These expansions of C-rich regions are present in a wide variety of organisms, and suggests that i-motifs could exist in vivo. It is postulated that i-motifs play roles in gene regulation and expression, telomerase inhibition, and DNA replication and repair. Although there are limited examples of i-motif formation in living cells, there are conditions that can be induced to create i-motifs. Coupling the examples of i-motif structures in cells with these experiments give avenues for further investigation.
Gene regulation and expression
Promoter regions of certain genes are C-rich. It is found in more than 40% of all human genes, especially in oncogenes, skeletal system development regions and areas of DNA processes, which strengthens the suggestion that i-motifs function as gene transcription regulators.[48][49][50] The promoter region of a transcription factor gene in silkworms, called BmPOUM2, was seen to form i-motif structures. The BmPOUM2 gene regulates another gene that affects wing disc cuticle formation during metamorphosis, and was seen to be positively regulated by i-motif formation.[12] This is an example of an important biological function in an organism being influenced by i-motif structure. Human telomeric DNA (hTelo) was also observed to form i-motif structures, also in vivo. This was confirmed by fluorescent marking with iMab.[51] These i-motif hTelos were found in regulatory regions of the human genome during the late G1 phase which indicates that i-motifs are involved in regulating genes important to development in the human genome. Even though more studies need to be conducted to validate these findings and provide specific insight to what genes are regulated, this study was important to opening the conversation of i-motif roles, and possible applications, in humans.
A similar role i-motifs can play is aiding the binding of transcription factors during gene transcription. One way this can occur is through temporary DNA unwinding into i-motif and g-quadruplex structures at promoter regions (like BCL2 ), which allows transcription of single strands of DNA.
Telomerase inhibition
The formation of g-quadruplexes and i-motifs at ends of chromosomes can lead to telomerase inhibition. The formation of i-motif structures at the ends of chromosomes inhibits telomerase from binding, which interferes with telomere lengthening. These formations result in the uncapping of telomeres, which exposes telomeres and triggers DNA damage response, ceasing rapid tumor growth.[52] Because i-motif structures are not specifically stable, the discovery of a ligand that selectively binds to i-motifs and stabilizes them was important to telomerase inhibition. Once bound with CSWNT, i-motifs were found to interfere with telomerase functions in vitro and in vivo in cancer cells, which was assessed by a TRAP assay.[53]
Ligand interaction
The binding of ligands can increase and modify i-motif functions. The first known selective ligand to bind with i-motif DNA is Carboxyl-modified single-walled carbon nanotubes (CSWNTs). These ligands binds to the 5' end major groove of DNA to induce i-motifs. The binding of CSWNT to i-motifs increases thermal stability at both acidic and biological pH by a significant amount. In this way, the CSWNT supports the formation of i-motif DNA over the Watson-Crick base pairing at pH 8.0.[54] Furthermore, many proteins and ligands fundamental to gene expression recognize C-rich oligonucleotides, such as Poly-C-binding protein (PCBP) and heterogeneous nuclear ribonucleoprotein K (HNRPK).
In the presence of C-rich single stranded oligonucleotides, PCBPs have the ability to play a variety of roles such as stabilizing mRNA and translational repression or enhancement depending on the C-rich single strand oligonucleotide that is being targeted.[55] Like PCBPs, the transcription factor heterogeneous nuclear ribonucleoprotein K (HNPRK) has the ability to selectively modulate the promoter regions of proteins such as KRAS and VGEF, in the presence of C-rich sequences such as i-motifs.[56][57] C-rich sequences such as i-motifs exist throughout the human genome, acting as targets for a variety of proteins that can regulate gene expression in multiple ways and locations.
DNA replication and repair
There was also evidence that i-motifs could interfere with DNA repair and replication. An experiment was performed where sequences that encouraged i-motif formation in a DNA strand that was being replicated by DNA polymerase. The focus of this experiment was the visualization of i-motifs in silkworms, and it was noted DNA polymerase was stalled, which implied i-motifs can impede DNA replication and repair.[58] The stalling effect of i-motif sequences was higher than hairpin DNA, although it is thermodynamically similar. This is due to the topology of i-motif DNA. The i-motif is unique when compared to other DNA because it is intercalated, which resists unwinding. This is what stalls DNA polymerase. It may also be attributed to steric hindrance, which would not allow DNA polymerase to bind.[16]
Other considerations
The formation of G-quadruplexes can lead its complementary DNA strand to be C-rich, which can form an i-motif, but this is not always the case. This is evident due to the majority of i-motif formation occurring in the G1 phase, while G-quadruplex formation is primarily noted in the S phase.
Applications
Applications of i-motifs are centered around biomedical topics, including bio-sensing, drug delivery systems, and molecular switches. Many of the current applications for i-motif DNA are due to its sensitivity to pH. The development of pH sensitive systems, which includes ligand binding, is a field of great interest to medicine, especially in the treatment and detection of cancer.
Bio-sensors
The conformational change from B DNA to i-motif under acidic conditions makes it useful as a colormetric sensor for glucose levels. A glucose detection system, Poly(24C)-MB, was created to detect a drop in pH levels in organisms, which occurs when glucose is oxidized. The dye of the Poly(24C)-MB system, methylene blue (MB), cannot bind when i-motifs are induced, giving rise to a color change that is easily visible. This system is simple, cost-effective, and precise due to i-motif conformation.[59]
Drug delivery systems
Gold nanoparticles/i-motif conjugated systems have been developed as a pH-induced drug delivery system. A study done using DNA conjugated gold nanoparticles (DNA-GNP) created a delivery molecule with stretches of C-rich single-stranded DNA that form i-motifs in cancer cell due to their acidic endosomes. When the DNA-GNP molecule enters a normal cell, no change takes place, but when the DNA-GNP enters a cancer cell, it induces i-motif conformation, which triggers doxorubicin (DOX), an effective cancer drug against leukemia and Hodgkin's lymphoma, to be released into the cell.[60] This method not only acts as an efficient drug delivery system, but can also be modified to detect cancer cells by including a dye or fluorescent, much like the colormetric sensor.
Theranostics
Due to i-motif formation in acidic conditions and cancer cells having acidic endosomes, cancer therapy and theranostic applications have been investigated. In a study by Takahashi et al., it was found that by using carboxyl-modified single-walled carbon nanotubes (C-SWNTs), telomerase activity could be inhibited, which could potentially lead to apoptosis of cancer cells. This is due to the use of fisetin, a plant flavanol, changing the conformation of i-motif structures into hairpin structures, which is a promising result in the investigation of various cancer drug therapies.[50] The binding of fisetin to an i-motif in the promoter region of vascular endothelial growth factor (VEGS), which is a signal protein for angiogenesis, induced a conformational change to a hairpin structure that inhibited it from functioning. The fisetin was suggested to bind to the loop of the i-motif, and when bound, it fluoresced. The fluorescent nature of this bond can be used as a diagnostic for this i-motif formation, and the formation of i-motifs that contain guanine residues. Overall, the study provided new information on how i-motifs can be used as a method for cancer treatment and detection.[61]
Molecular switches
A study at the University of Bonn explained how i-motifs can be utilized as molecular switches. The study synthesized a ring of DNA with certain regions of C-rich DNA. At a pH of 5, these regions contracted to form i-motifs, tightening the ring in a fashion similar to closing a trash bag. At a pH of 8 the i-motif regions collapsed back into their linear forms, relaxing the ring. DNA rings that can tighten and loosen based on pH can be used to build more complex structures of interlocking DNA like catenanes and rotaxanes.[62] This study emphasized that the manipulation of i-motif structure can unlock new possibilities in nanomechanics. Another study showed CSWNTs could induce i-motif formation in human telomeric DNA and modify it by attaching a redox active methylene blue group to the 3' end and an electrode to the 5' end. In the i-motif conformation this modified DNA strand produces a large increase in Faradaic current, which only reacts to CSWNTs, allowing researchers to detect a specific type of carbon nanotube with a direct detection limit of 0.2 ppm.
See also
References
- ↑ Gehring, Kalle; Leroy, Jean-Louis; Guéron, Maurice (June 1993). "A tetrameric DNA structure with protonated cytosine-cytosine base pairs". Nature. 363 (6429): 561–565. Bibcode:1993Natur.363..561G. doi:10.1038/363561a0. ISSN 0028-0836. PMID 8389423. S2CID 388606.
- 1 2 Zeraati, Mahdi; Langley, David B.; Schofield, Peter; Moye, Aaron L.; Rouet, Romain; Hughes, William E.; Bryan, Tracy M.; Dinger, Marcel E.; Christ, Daniel (June 2018). "I-motif DNA structures are formed in the nuclei of human cells". Nature Chemistry. 10 (6): 631–637. Bibcode:2018NatCh..10..631Z. doi:10.1038/s41557-018-0046-3. ISSN 1755-4330. PMID 29686376. S2CID 13816298.
- ↑ Chaudhary, Swati; Kaushik, Mahima; Ahmed, Saami; Kukreti, Shrikant (7 December 2020). "Exploring potential of i-motif DNA formed in the promoter region of GRIN1 gene for nanotechnological applications". Results in Chemistry. 2: 100086. doi:10.1016/j.rechem.2020.100086. ISSN 2211-7156.
- ↑ SCIMEX (23 April 2018). "Found: a new form of DNA in our cells". Scimex. Retrieved 10 December 2020.
- ↑ Benabou, S.; Aviñó, A.; Eritja, R.; González, C.; Gargallo, R. (2014). "Fundamental aspects of the nucleic acid i-motif structures". RSC Advances. 4 (51): 26956–26980. Bibcode:2014RSCAd...426956B. doi:10.1039/C4RA02129K. hdl:10261/126489. ISSN 2046-2069.
- ↑ Snoussi, K.; Nonin-Lecomte, S.; Lerou, J.L. (30 May 2001). "THE RNA I-MOTIF". Journal of Molecular Biology. 309 (1): 139–53. doi:10.2210/pdb1i9k/pdb. PMID 11491284.
- ↑ Gurung, Sarah P.; Schwarz, Christine; Hall, James P.; Cardin, Christine J.; Brazier, John A. (2015). "The importance of loop length on the stability of i-motif structures". Chemical Communications. 51 (26): 5630–5632. doi:10.1039/c4cc07279k. ISSN 1359-7345. PMC 4384421. PMID 25686374.
- ↑ Benabou, S.; Aviñó, A.; Eritja, R.; González, C.; Gargallo, R. (2014). "Fundamental aspects of the nucleic acid i-motif structures" (PDF). RSC Advances. 4 (51): 26956–26980. Bibcode:2014RSCAd...426956B. doi:10.1039/c4ra02129k. hdl:10261/126489. ISSN 2046-2069.
- ↑ Abou Assi, Hala; Garavís, Miguel; González, Carlos; Damha, Masad J (19 September 2018). "i-Motif DNA: structural features and significance to cell biology". Nucleic Acids Research. 46 (16): 8038–8056. doi:10.1093/nar/gky735. ISSN 0305-1048. PMC 6144788. PMID 30124962.
- ↑ Malliavin, Thérèse E.; Gau, Jocelyne; Snoussi, Karim; Leroy, Jean-Louis (June 2003). "Stability of the I-motif Structure Is Related to the Interactions between Phosphodiester Backbones". Biophysical Journal. 84 (6): 3838–3847. Bibcode:2003BpJ....84.3838M. doi:10.1016/S0006-3495(03)75111-X. ISSN 0006-3495. PMC 1302965. PMID 12770889.
- 1 2 Dettler, Jamie M.; Buscaglia, Robert; Cui, JingJing; Cashman, Derek; Blynn, Meredith; Lewis, Edwin A. (July 2010). "Biophysical Characterization of an Ensemble of Intramolecular i-Motifs Formed by the Human c-MYC NHE III1 P1 Promoter Mutant Sequence". Biophysical Journal. 99 (2): 561–567. Bibcode:2010BpJ....99..561D. doi:10.1016/j.bpj.2010.04.042. ISSN 0006-3495. PMC 2905117. PMID 20643075.
- 1 2 Wright, Elisé P.; Huppert, Julian L.; Waller, Zoë A. E. (9 February 2017). "Identification of multiple genomic DNA sequences which form i-motif structures at neutral pH". Nucleic Acids Research. 45 (6): 2951–2959. doi:10.1093/nar/gkx090. ISSN 0305-1048. PMC 5605235. PMID 28180276.
- ↑ Sun, Daekyu; Hurley, Laurence H. (14 May 2009). "The importance of negative superhelicity in inducing the formation of G-quadruplex and i-motif structures in the c-Myc promoter: implications for drug targeting and control of gene expression". Journal of Medicinal Chemistry. 52 (9): 2863–2874. doi:10.1021/jm900055s. ISSN 0022-2623. PMC 2757002. PMID 19385599.
- ↑ Pandit, Sarwar Ahmad; Rather, Mudasir Ahmad; Bhat, Sajad Ahmad; Jan, Roohi; Rather, Ghulam Mohd; Bhat, Mohsin Ahmad (21 June 2017). "An Insight into a Fascinating DMF-Water Mixed Solvent System: Physicochemical and Electrochemical Studies". ChemistrySelect. 2 (18): 5115–5127. doi:10.1002/slct.201700553. ISSN 2365-6549.
- 1 2 Malliavin, Thérèse E.; Gau, Jocelyne; Snoussi, Karim; Leroy, Jean-Louis (June 2003). "Stability of the I-motif Structure Is Related to the Interactions between Phosphodiester Backbones". Biophysical Journal. 84 (6): 3838–3847. Bibcode:2003BpJ....84.3838M. doi:10.1016/s0006-3495(03)75111-x. ISSN 0006-3495. PMC 1302965. PMID 12770889.
- 1 2 Abou Assi, Hala; Garavís, Miguel; González, Carlos; Damha, Masad J (16 August 2018). "i-Motif DNA: structural features and significance to cell biology". Nucleic Acids Research. 46 (16): 8038–8056. doi:10.1093/nar/gky735. ISSN 0305-1048. PMC 6144788. PMID 30124962.
- ↑ Yang, Bo; Rodgers, M. T. (19 December 2013). "Base-Pairing Energies of Proton-Bound Heterodimers of Cytosine and Modified Cytosines: Implications for the Stability of DNAi-Motif Conformations". Journal of the American Chemical Society. 136 (1): 282–290. doi:10.1021/ja409515v. ISSN 0002-7863. PMID 24320604.
- ↑ Lieblein, Anna Lena; Krämer, Maximilian; Dreuw, Andreas; Fürtig, Boris; Schwalbe, Harald (12 March 2012). "The Nature of Hydrogen Bonds in Cytidine⋅⋅⋅H+⋅⋅⋅Cytidine DNA Base Pairs". Angewandte Chemie International Edition. 51 (17): 4067–4070. doi:10.1002/anie.201200549. ISSN 1433-7851. PMID 22411471.
- 1 2 Leroy, Jean Louis; Gehring, Kalle; Kettani, Abdelali; Gueron, Maurice (15 June 1993). "Acid multimers of oligodeoxycytidine strands: Stoichiometry, base-pair characterization, and proton exchange properties". Biochemistry. 32 (23): 6019–6031. doi:10.1021/bi00074a013. ISSN 0006-2960. PMID 8389586.
- 1 2 3 Peng, Yinghua; Wang, Xiaohui; Xiao, Yi; Feng, Lingyan; Zhao, Chao; Ren, Jinsong; Qu, Xiaogang (30 September 2009). "i-Motif Quadruplex DNA-Based Biosensor for Distinguishing Single- and Multiwalled Carbon Nanotubes". Journal of the American Chemical Society. 131 (38): 13813–13818. doi:10.1021/ja9051763. ISSN 0002-7863. PMID 19736925.
- 1 2 Wright, Elisé P.; Lamparska, Katarzyna; Smith, Steven S.; Waller, Zoë A. E. (30 August 2017). "Substitution of Cytosine with Guanylurea Decreases the Stability of i-Motif DNA". Biochemistry. 56 (36): 4879–4883. doi:10.1021/acs.biochem.7b00628. ISSN 0006-2960. PMID 28853563.
- ↑ Mir, B.; Solés, X.; González, C.; Escaja, N. (5 June 2017). "The effect of the neutral cytidine protonated analogue pseudoisocytidine on the stability of i-motif structures". Scientific Reports. 7 (1): 2772. Bibcode:2017NatSR...7.2772M. doi:10.1038/s41598-017-02723-y. ISSN 2045-2322. PMC 5459817. PMID 28584239.
- 1 2 3 Bhavsar-Jog, Yogini P.; Van Dornshuld, Eric; Brooks, Tracy A.; Tschumper, Gregory S.; Wadkins, Randy M. (6 March 2014). "Epigenetic Modification, Dehydration, and Molecular Crowding Effects on the Thermodynamics of i-Motif Structure Formation from C-Rich DNA". Biochemistry. 53 (10): 1586–1594. doi:10.1021/bi401523b. ISSN 0006-2960. PMC 3985701. PMID 24564458.
- ↑ Lannes, Laurie; Halder, Saheli; Krishnan, Yamuna; Schwalbe, Harald (30 June 2015). "Tuning the pH Response of i-Motif DNA Oligonucleotides". ChemBioChem. 16 (11): 1647–1656. doi:10.1002/cbic.201500182. ISSN 1439-4227. PMID 26032298. S2CID 28930880.
- ↑ Xu, Baochang; Devi, Gitali; Shao, Fangwei (2015). "Regulation of telomeric i-motif stability by 5-methylcytosine and 5-hydroxymethylcytosine modification". Organic & Biomolecular Chemistry. 13 (20): 5646–5651. doi:10.1039/c4ob02646b. ISSN 1477-0520. PMID 25886653.
- ↑ Kanehara, Hideyuki; Mizuguchi, Masatsugu; Tajima, Kunihiko; Kanaori, Kenji; Makino, Keisuke (February 1997). "Spectroscopic Evidence for the Formation of Four-Stranded Solution Structure of Oligodeoxycytidine Phosphorothioate†". Biochemistry. 36 (7): 1790–1797. doi:10.1021/bi961528c. ISSN 0006-2960. PMID 9048563.
- 1 2 Mergny, J. (1 November 1998). "Kinetics and thermodynamics of i-DNA formation: phosphodiester versus modified oligodeoxynucleotides". Nucleic Acids Research. 26 (21): 4797–4803. doi:10.1093/nar/26.21.4797. ISSN 1362-4962. PMC 147917. PMID 9776737.
- 1 2 3 Day, Henry A.; Pavlou, Pavlos; Waller, Zoë A.E. (August 2014). "i-Motif DNA: Structure, stability and targeting with ligands". Bioorganic & Medicinal Chemistry. 22 (16): 4407–4418. doi:10.1016/j.bmc.2014.05.047. ISSN 0968-0896. PMID 24957878.
- ↑ Miyoshi, Daisuke; Matsumura, Shizuka; Nakano, Shu-ichi; Sugimoto, Naoki (January 2004). "Duplex Dissociation of Telomere DNAs Induced by Molecular Crowding". Journal of the American Chemical Society. 126 (1): 165–169. doi:10.1021/ja036721q. ISSN 0002-7863. PMID 14709080.
- ↑ Cui, Jingjing; Waltman, Phillip; Le, Vu; Lewis, Edwin (15 October 2013). "The Effect of Molecular Crowding on the Stability of Human c-MYC Promoter Sequence I-Motif at Neutral pH". Molecules. 18 (10): 12751–12767. doi:10.3390/molecules181012751. ISSN 1420-3049. PMC 6270392. PMID 24132198.
- 1 2 Sun, Daekyu; Hurley, Laurence H. (14 May 2009). "The Importance of Negative Superhelicity in Inducing the Formation of G-Quadruplex and i-Motif Structures in the c-Myc Promoter: Implications for Drug Targeting and Control of Gene Expression". Journal of Medicinal Chemistry. 52 (9): 2863–2874. doi:10.1021/jm900055s. ISSN 0022-2623. PMC 2757002. PMID 19385599.
- ↑ Krishnan-Ghosh, Yamuna; Stephens, Elaine; Balasubramanian, Shankar (2005). "PNA forms an i-motif". Chemical Communications (42): 5278–80. doi:10.1039/b510405j. ISSN 1359-7345. PMID 16244727.
- ↑ Saxena, Sarika; Bansal, Aparna; Kukreti, Shrikant (March 2008). "Structural polymorphism exhibited by a homopurine·homopyrimidine sequence found at the right end of human c-jun protooncogene". Archives of Biochemistry and Biophysics. 471 (2): 95–108. doi:10.1016/j.abb.2008.01.015. ISSN 0003-9861. PMID 18262488.
- ↑ Benabou, Sanae; Ferreira, Rubén; Aviñó, Anna; González, Carlos; Lyonnais, Sébastien; Solà, Maria; Eritja, Ramon; Jaumot, Joaquim; Gargallo, Raimundo (January 2014). "Solution equilibria of cytosine- and guanine-rich sequences near the promoter region of the n-myc gene that contain stable hairpins within lateral loops". Biochimica et Biophysica Acta (BBA) - General Subjects. 1840 (1): 41–52. doi:10.1016/j.bbagen.2013.08.028. hdl:2445/46204. ISSN 0304-4165. PMID 24012973.
- 1 2 Lacroix, Laurent; Mergny, Jean-Louis (September 2000). "Chemical Modification of Pyrimidine TFOs: Effect on i-Motif and Triple Helix Formation". Archives of Biochemistry and Biophysics. 381 (1): 153–163. doi:10.1006/abbi.2000.1934. ISSN 0003-9861. PMID 11019831.
- 1 2 Zeraati, Mahdi; Langley, David B.; Schofield, Peter; Moye, Aaron L.; Rouet, Romain; Hughes, William E.; Bryan, Tracy M.; Dinger, Marcel E.; Christ, Daniel (23 April 2018). "I-motif DNA structures are formed in the nuclei of human cells". Nature Chemistry. 10 (6): 631–637. Bibcode:2018NatCh..10..631Z. doi:10.1038/s41557-018-0046-3. ISSN 1755-4330. PMID 29686376. S2CID 13816298.
- ↑ Kang, Hyun-Jin; Kendrick, Samantha; Hecht, Sidney M.; Hurley, Laurence H. (7 March 2014). "The Transcriptional Complex Between theBCL2i-Motif and hnRNP LL Is a Molecular Switch for Control of Gene Expression That Can Be Modulated by Small Molecules". Journal of the American Chemical Society. 136 (11): 4172–4185. doi:10.1021/ja4109352. ISSN 0002-7863. PMC 3985447. PMID 24559432.
- ↑ Zhang, Xi Yuan; Luo, Hong Qun; Li, Nian Bing (15 June 2014). "Crystal violet as an i-motif structure probe for reversible and label-free pH-driven electrochemical switch". Analytical Biochemistry. 455: 55–59. doi:10.1016/j.ab.2014.03.015. ISSN 0003-2697. PMID 24699211.
- ↑ Warner, S.L.; Vankayalapati, H.; Bashyam, S.; Grand, C.L.; Han, H.; Von Hoff, D.D.; Hurley, L.H.; Bearss, D.J. (September 2004). "126 Development of a new series of tricyclic pyrimido-indole inhibitors targeting Aurora kinases". European Journal of Cancer Supplements. 2 (8): 41. doi:10.1016/s1359-6349(04)80134-4. ISSN 1359-6349.
- ↑ Siddiqui-Jain, A.; Grand, C. L.; Bearss, D. J.; Hurley, L. H. (23 August 2002). "Direct evidence for a G-quadruplex in a promoter region and its targeting with a small molecule to repress c-MYC transcription". Proceedings of the National Academy of Sciences. 99 (18): 11593–11598. Bibcode:2002PNAS...9911593S. doi:10.1073/pnas.182256799. ISSN 0027-8424. PMC 129314. PMID 12195017.
- ↑ Fernández, Sergio; Eritja, Ramon; Aviñó, Anna; Jaumot, Joaquim; Gargallo, Raimundo (November 2011). "Influence of pH, temperature and the cationic porphyrin TMPyP4 on the stability of the i-motif formed by the 5'-(C3TA2)4-3' sequence of the human telomere". International Journal of Biological Macromolecules. 49 (4): 729–736. doi:10.1016/j.ijbiomac.2011.07.004. hdl:10261/47689. ISSN 0141-8130. PMID 21777611.
- ↑ Neidle, Stephen (2012), "RNA Quadruplexes", Therapeutic Applications of Quadruplex Nucleic Acids, Elsevier, pp. 139–149, doi:10.1016/b978-0-12-375138-6.00008-x, ISBN 978-0-12-375138-6, retrieved 16 December 2020
- 1 2 3 4 5 Sedghi Masoud, Shadi; Nagasawa, Kazuo (1 December 2018). "i-Motif-Binding Ligands and Their Effects on the Structure and Biological Functions of i-Motif". Chemical and Pharmaceutical Bulletin. 66 (12): 1091–1103. doi:10.1248/cpb.c18-00720. ISSN 0009-2363. PMID 30504626.
- ↑ Ni, Nan; Qu, Bo; Tian, Di; Cong, Zhiyuan; Wang, Weiping; Gao, Chao; Xiao, Lixin; Chen, Zhijian; Gong, Qihuang; Wei, Wei (2 May 2013). "Macromol. Chem. Phys. 9/2013". Macromolecular Chemistry and Physics. 214 (9): 961. doi:10.1002/macp.201370030. ISSN 1022-1352.
- ↑ Nagasawa, Kazuo; Sedghi Masoud, Shadi; Tsushima, Yamato; Iida, Keisuke (2015). "Synthesis of Macrocyclic Penta- and Tetraoxazoles as G-Quadruplex Ligands". Heterocycles. 90 (2): 866. doi:10.3987/com-14-s(k)90. ISSN 0385-5414.
- ↑ Sheng, Qiran; Neaverson, Joseph C.; Mahmoud, Tasnim; Stevenson, Clare E. M.; Matthews, Susan E.; Waller, Zoë A. E. (2017). "Identification of new DNA i-motif binding ligands through a fluorescent intercalator displacement assay". Organic & Biomolecular Chemistry. 15 (27): 5669–5673. doi:10.1039/c7ob00710h. ISSN 1477-0520. PMC 5708337. PMID 28567459.
- ↑ Li, X.; Peng, Y.; Ren, J.; Qu, X. (13 December 2006). "Carboxyl-modified single-walled carbon nanotubes selectively induce human telomeric i-motif formation". Proceedings of the National Academy of Sciences. 103 (52): 19658–19663. Bibcode:2006PNAS..10319658L. doi:10.1073/pnas.0607245103. ISSN 0027-8424. PMC 1750900. PMID 17167055.
- ↑ Fleming, Aaron M.; Ding, Yun; Rogers, R. Aaron; Zhu, Judy; Zhu, Julia; Burton, Ashlee D.; Carlisle, Connor B.; Burrows, Cynthia J. (5 April 2017). "4n–1 Is a "Sweet Spot" in DNA i-Motif Folding of 2'-Deoxycytidine Homopolymers". Journal of the American Chemical Society. 139 (13): 4682–4689. doi:10.1021/jacs.6b10117. ISSN 0002-7863. PMID 28290680.
- ↑ Wright, Elisé P.; Huppert, Julian L.; Waller, Zöe A.E. (13 November 2017). "Identification of multiple genomic DNA sequences which form i-motif structures at neutral pH". Nucleic Acids Research. 45 (22): 13095–13096. doi:10.1093/nar/gkx1178. ISSN 0305-1048. PMC 5728391. PMID 29140476.
- 1 2 Takahashi, Shuntaro; Bhattacharjee, Snehasish; Ghosh, Saptarshi; Sugimoto, Naoki; Bhowmik, Sudipta (December 2020). "Preferential targeting cancer-related i-motif DNAs by the plant flavonol fisetin for theranostics applications". Scientific Reports. 10 (1): 2504. Bibcode:2020NatSR..10.2504T. doi:10.1038/s41598-020-59343-2. ISSN 2045-2322. PMC 7018961. PMID 32054927.
- ↑ Zeraati, Mahdi; Langley, David B.; Schofield, Peter; Moye, Aaron L.; Rouet, Romain; Hughes, William E.; Bryan, Tracy M.; Dinger, Marcel E.; Christ, Daniel (2018). "I-motif DNA structures are formed in the nuclei of human cells". Nature Chemistry. 10 (6): 631–637. Bibcode:2018NatCh..10..631Z. doi:10.1038/s41557-018-0046-3. PMID 29686376. S2CID 13816298.
- ↑ Amato, Jussara; Iaccarino, Nunzia; Randazzo, Antonio; Novellino, Ettore; Pagano, Bruno (24 June 2014). "Noncanonical DNA Secondary Structures as Drug Targets: the Prospect of the i-Motif". ChemMedChem. 9 (9): 2026–2030. doi:10.1002/cmdc.201402153. ISSN 1860-7179. PMID 24962454. S2CID 39651503.
- ↑ Chen, Yong; Qu, Konggang; Zhao, Chuanqi; Wu, Li; Ren, Jinsong; Wang, Jiasi; Qu, Xiaogang (January 2012). "Insights into the biomedical effects of carboxylated single-wall carbon nanotubes on telomerase and telomeres". Nature Communications. 3 (1): 1074. Bibcode:2012NatCo...3.1074C. doi:10.1038/ncomms2091. ISSN 2041-1723. PMID 23011128.
- ↑ Sushmita, Nautiyal (June 2020). "I-MOTIF DNA: SIGNIFICANCE AND FUTURE PROSPECTIVE".
- ↑ Yoga, Y. M. K.; Traore, D. A. K.; Sidiqi, M.; Szeto, C.; Pendini, N. R.; Barker, A.; Leedman, P. J.; Wilce, J. A.; Wilce, M. C. J. (1 June 2012). "Contribution of the first K-homology domain of poly(C)-binding protein 1 to its affinity and specificity for C-rich oligonucleotides". Nucleic Acids Research. 40 (11): 5101–5114. doi:10.1093/nar/gks058. ISSN 0305-1048. PMC 3367169. PMID 22344691.
- ↑ Kaiser, Christine E.; Van Ert, Natalie A.; Agrawal, Prashansa; Chawla, Reena; Yang, Danzhou; Hurley, Laurence H. (28 June 2017). "Insight into the Complexity of the i-Motif and G-Quadruplex DNA Structures Formed in the KRAS Promoter and Subsequent Drug-Induced Gene Repression". Journal of the American Chemical Society. 139 (25): 8522–8536. doi:10.1021/jacs.7b02046. ISSN 0002-7863. PMC 5978000. PMID 28570076.
- ↑ Uribe, Diana J.; Guo, Kexiao; Shin, Yoon-Joo; Sun, Daekyu (10 May 2011). "Heterogeneous Nuclear Ribonucleoprotein K and Nucleolin as Transcriptional Activators of the Vascular Endothelial Growth Factor Promoter through Interaction with Secondary DNA Structures". Biochemistry. 50 (18): 3796–3806. doi:10.1021/bi101633b. ISSN 0006-2960. PMC 3119528. PMID 21466159.
- ↑ Tang, Wenhuan; Niu, Kangkang; Yu, Guoxing; Jin, Ying; Zhang, Xian; Peng, Yuling; Chen, Shuna; Deng, Huimin; Li, Sheng; Wang, Jian; Song, Qisheng (5 March 2020). "In vivo visualization of the i-motif DNA secondary structure in the Bombyx mori testis". Epigenetics & Chromatin. 13 (1): 12. doi:10.1186/s13072-020-00334-y. ISSN 1756-8935. PMC 7059380. PMID 32138783.
- ↑ Wang, Qin; Dai, Tianyue; Sun, Pengfei; Wang, Xiayan; Wang, Guangfeng (1 August 2020). "A colorimetric and ratiometric glucose sensor based on conformational switch of i-motif DNA". Talanta Open. 1: 100001. doi:10.1016/j.talo.2020.100001. ISSN 2666-8319.
- ↑ Song, Lei; Ho, Vincent H. B.; Chen, Chun; Yang, Zhongqiang; Liu, Dongsheng; Chen, Rongjun; Zhou, Dejian (2013). "Drug Delivery: Efficient, pH-Triggered Drug Delivery Using a pH-Responsive DNA-Conjugated Gold Nanoparticle (Adv. Healthcare Mater. 2/2013)". Advanced Healthcare Materials. 2 (2): 380. doi:10.1002/adhm.201370008. ISSN 2192-2659.
- ↑ Takahashi, Shuntaro; Bhattacharjee, Snehasish; Ghosh, Saptarshi; Sugimoto, Naoki; Bhowmik, Sudipta (13 February 2020). "Preferential targeting cancer-related i-motif DNAs by the plant flavonol fisetin for theranostics applications". Scientific Reports. 10 (1): 2504. Bibcode:2020NatSR..10.2504T. doi:10.1038/s41598-020-59343-2. ISSN 2045-2322. PMC 7018961. PMID 32054927.
- ↑ Li, Tao; Famulok, Michael (22 January 2013). "I-Motif-Programmed Functionalization of DNA Nanocircles". Journal of the American Chemical Society. 135 (4): 1593–1599. doi:10.1021/ja3118224. ISSN 0002-7863. PMID 23312021.