 R = H or CH3C(=O) | |
| Names | |
|---|---|
| Other names
glycerol acetate glyceryl acetate 1,2,3-propanetriol acetate | |
| Identifiers | |
| |
3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.014.216 |
| EC Number |
|
PubChem CID |
|
| RTECS number |
|
| UNII |
|
| |
| |
| Properties | |
| Variable | |
| Molar mass | Variable |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
Glycerine acetate is a mixture of esters produced from the esterification of glycerol (1) with acetic acid. This reaction produces five congeners:
- the two monoacetylglycerols / MAG / monoacetin (2 and 3)
- the two diacetylglycerols / DAG / diacetin / glyceryl diacetate (4 and 5)
- the one triacetalglycerol / TAG / triacetin (6)
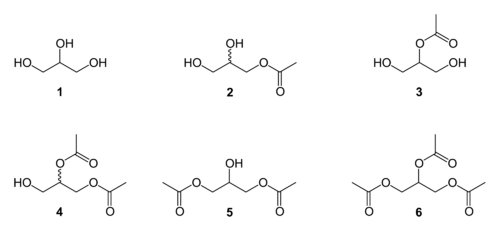
Structures of all possible acetate esters of glycerol
DAG and TAG can be used as fuel additives for improving the cold and viscosity properties of biodiesel or the antiknocking properties of gasoline.[1]
Notes
- ↑ J. A. Melero; R. vanGrieken; G. Morales; M. Paniagua (2007). "Acidic Mesoporous Silica for the Acetylation of Glycerol: Synthesis of Bioadditives to Petrol Fuel". Energy & Fuels. 21 (3): 1782–1791. doi:10.1021/ef060647q.
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.