 | |
| Names | |
|---|---|
| Preferred IUPAC name
1,3-Diphenylpropane-1,3-dione | |
| Other names
2-Benzoylacetophenone | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.003.999 |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C15H12O2 | |
| Molar mass | 224.25 g/mol |
| Appearance | white solid |
| Density | 1.334 g/cm3 |
| Melting point | 77 to 78 °C (171 to 172 °F; 350 to 351 K)[1] |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
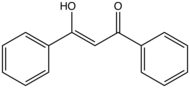
Dibenzoylmethane (DBM) is an organic compound with the formula (C6H5C(O))2CH2. DBM is the name for a 1,3-diketone, but the compound exists primarily as one of two equivalent enol tautomers.[2] DBM (actually its enol) is a white solid. Due to their high photostability and UV-absorbing properties, derivatives of DBM such as avobenzone, have found applications as sunscreen products.
Synthesis and reactions
DBM is prepared by condensation of ethyl benzoate with acetophenone.[3]
Like other 1,3-diketones (or their enols), DBM condenses with a variety of bifunctional reagents to give heterocycles. Hydrazine gives diphenylpyrazole. Urea and thiourea also condense to give six-membered rings. With metal salts, the conjugate base of DBM forms complexes akin to the metal acetylacetonates.
Occurrence and medicinal properties
Dibenzoylmethane (DBM) is a minor constituent in the root extract of Licorice (Glycyrrhiza glabra in the family Leguminosae).[4] It is also found in Curcumin. These occurrences have led to investigations into the medicinal properties of this class of compounds.[5][6]
DBM (and Trazodone) slow disease progression by preventing the cessation of protein synthesis in neurons.[7]
Related compounds
References
- ↑ Jan Zawadiak; Marek Mrzyczek (2010). "UV absorption and keto–enol tautomerism equilibrium of methoxy and dimethoxy 1,3-diphenylpropane-1,3-diones". Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy. 75 (2): 925–929. Bibcode:2010AcSpA..75..925Z. doi:10.1016/j.saa.2009.12.040. PMID 20047853.
- ↑ Thomas, L. H.; Florence, A. J.; Wilson, C. C. (2009). "Hydrogen atom behaviour imaged in a short intramolecular hydrogen bond using the combined approach of X-ray and neutron diffraction". New Journal of Chemistry. 33 (12): 2486–2490. doi:10.1039/B908915B.
- ↑ Magnani, Arthur; McElvain, S. M. (1940). "Dibenzoylmethane". Org. Synth. 20: 32. doi:10.15227/orgsyn.020.0032.
- ↑ Jackson, Kimberly M.; Deleon, Marisela; Verret, C.Reynold; Harris, Wayne B. (2002). "Dibenzoylmethane Induces Cell Cycle Deregulation in Human Prostate Cancer Cells". Cancer Letters. 178 (2): 161–165. doi:10.1016/S0304-3835(01)00844-8. PMID 11867200.
- ↑ Shishu; Singla, A.K.; Kaur, I.P. (2003). "Inhibitory Effect of Dibenzoylmethane on Mutagenicity of Food-Derived Heterocyclic Amine Mutagens". Phytomedicine. 10 (6–7): 575–582. doi:10.1078/094471103322331575. PMID 13678246.
- ↑ Pan, Min-Hsiung; Huang, Mei-Chen; Wang, Ying-Jan; Lin, Jen-Kun; Lin, Chao-Hsien (2003). "Induction of Apoptosis by Hydroxydibenzoylmethane through Coordinative Modulation of Cyclin D3, BCL-XL, and Bax, Release of Cytochrome c, and Sequential Activation of Caspases in Human Colorectal Carcinoma Cells". Journal of Agricultural and Food Chemistry. 51 (14): 3977–3984. doi:10.1021/jf034094i. PMID 12822933.
- ↑ Halliday, Mark; Radford, Helois; Zents, Karlijn A. M.; Molloy, Collin; Moreno, Julie A.; Verity, Nicholas C.; Smith, Ewan; Ortori, Catharine A.; Barrett, David A.; Bushell, Martin; Mallucci, Giovanna R. (2017). "Repurposed Drugs Targeting eIF2α-P-Mediated Translational Repression Prevent Neurodegeneration in Mice". Brain. 140 (6): 1768–1783. doi:10.1093/brain/awx074. PMC 5445255. PMID 28430857.
