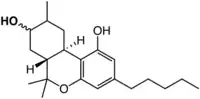
 | |
| Names | |
|---|---|
| Other names
8-OH-HHC | |
| Identifiers | |
3D model (JSmol) |
|
PubChem CID |
|
| |
| Properties | |
| C21H32O3 | |
| Molar mass | 332.484 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
8-Hydroxyhexahydrocannabinols (8-OH-9α-HHC and 8-OH-9β-HHC) are active primary metabolites of hexahydrocannabinol (HHC) in animals and trace phytocannabinoids. The 8-OH-HHCs are produced in notable concentrations following HHC administration in several animal species, including humans. They have drawn research interest for their role in HHC toxicology and stereoisomeric probes of the cannabinoid drug/receptor interaction.
Like Δ9-THC and Δ8-THC, HHC is processed by cytochrome p450 (CYP3A4, CYP2C9 and CYP2C19) to a series of oxygenated derivatives, some of which maintain activity.[1] While 11-OH-HHC and its downstream products are the major metabolites of HHC metabolism, hydroxylation at C8 plays a varyingly significant role in animal species.[2][3] Metabolite ratios are also subject to interspecies variation, with one study finding mice hepatocytes preferentially produced 8α-OH-HHC (49/5 α/β) while hamster hematocytes evidenced the opposing selectivity (20/43 α/β).[2]
While 11-OH-HHC is quickly oxidized to the inactive, water-soluble 11-COOH-HHC, further oxidation of 8-OH instead yields the 8-oxo derivatives, which are then conjugated and excreted.[4]
Stereoisomerism

There are four possible 8-OH-HHC metabolites arising from naturally derived HHCs: cis- and trans-8-OH-9α-HHC & cis- and trans-8-OH-9β-HHC. All four have been prepared synthetically to probe stereochemical effects on cannabinoid biological activity.[5] In in vivo tests on rhesus macaques, Mechoulam and coworkers found the highest activity in the cis-8-OH-9β-HHC stereoisomer.[6] All four forms are believed to be active.
References
- ↑ Huestis, Marilyn A. (August 2007). "Human Cannabinoid Pharmacokinetics". Chemistry & Biodiversity. 4 (8): 1770–1804. doi:10.1002/cbdv.200790152. PMC 2689518. PMID 17712819.
- 1 2 Harvey, D.J.; Brown, N.K. (November 1991). "Comparative in vitro metabolism of the cannabinoids". Pharmacology Biochemistry and Behavior. 40 (3): 533–540. doi:10.1016/0091-3057(91)90359-A. PMID 1806943. S2CID 25827210.
- ↑ Dinis-Oliveira, Ricardo Jorge (2016-01-02). "Metabolomics of Δ 9 -tetrahydrocannabinol: implications in toxicity". Drug Metabolism Reviews. 48 (1): 80–87. doi:10.3109/03602532.2015.1137307. ISSN 0360-2532. PMID 26828228. S2CID 32913023.
- ↑ Lewis, John; Molnar, Anna; Allsop, David; Copeland, Jan; Fu, Shanlin (January 2016). "Rapid elimination of Carboxy-THC in a cohort of chronic cannabis users". International Journal of Legal Medicine. 130 (1): 147–152. doi:10.1007/s00414-015-1241-z. ISSN 0937-9827. PMID 26233612. S2CID 25318521.
- ↑ Mechoulam, R.; Lander, N.; Varkony, T. H.; Kimmel, I.; Becker, O.; Ben-Zvi, Z.; Edery, H.; Porath, G. (October 1980). "Stereochemical requirements for cannabinoid activity". Journal of Medicinal Chemistry. 23 (10): 1068–1072. doi:10.1021/jm00184a002. ISSN 0022-2623. PMID 7420350.
- ↑ Harvey, D. J.; Brown, N. K. (1991-05-01). "In vitro metabolism of the equatorial C11-methyl isomer of hexahydrocannabinol in several mammalian species". Drug Metabolism and Disposition. 19 (3): 714–716. ISSN 0090-9556. PMID 1680642.