 | |
 | |
| Names | |
|---|---|
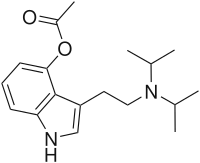
| Preferred IUPAC name
3-{2-[Di(propan-2-yl)amino]ethyl}-1H-indol-4-yl acetate | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C18H26N2O2 | |
| Molar mass | 302.418 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
4-Acetoxy-DiPT (4-acetoxy-N,N-diisopropyltryptamine, ipracetin) is a synthetic psychedelic tryptamine. It is relatively uncommon and has only a short history of human use.
Chemistry
4-AcO-DiPT is a tryptamine structurally similar to 4-HO-DiPT and psilocin.
Drug prohibition laws
Denmark
4-AcO-DiPT is added to the list of Schedule B controlled substances.[1]
Japan
4-Acetoxy-DiPT is a controlled substance in Japan.[2]
Sweden
Sveriges riksdags health ministry Statens folkhälsoinstitut classified 4-AcO-DiPT as "health hazard" under the act Lagen om förbud mot vissa hälsofarliga varor (translated Act on the Prohibition of Certain Goods Dangerous to Health) as of Mar 1, 2005, in their regulation SFS 2005:26 listed as 4-acetoxi-N,N-diisopropyltryptamin (4-AcO-DIPT), making it illegal to sell or possess.[3]
United States
4-Acetoxy-DiPT is an unscheduled substance in the United States. Due to similarities to other scheduled tryptamines such as psilocin and DiPT, possession may be prosecuted under the Federal Analog Act in the United States.
See also
References
- ↑ "Bekendtgørelse om euforiserende stoffer - retsinformation.dk".
- ↑ "指定薬物一覧|厚生労働省". Retrieved 23 December 2011.
- ↑ "1Svensk författningssamling" (PDF). www.notisum.se. 3 February 2005. Archived from the original (PDF) on 2013-09-29. Retrieved 2020-03-24.