 | |
 | |
| Names | |
|---|---|
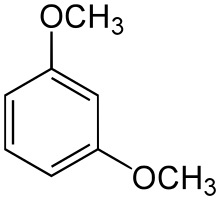
| IUPAC name
1,3-Dimethoxybenzene | |
| Other names
Dimethylresorcinol Resorcinol dimethyl ether | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.005.259 |
| EC Number |
|
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C8H10O2 | |
| Molar mass | 138.166 g·mol−1 |
| Related compounds | |
Related compounds |
1,2-Dimethoxybenzene; 1,4-Dimethoxybenzene |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
1,3-Dimethoxybenzene is an organic compound with the formula C6H4(OCH3)2. It is one of three isomers of dimethoxybenzene.
Uses
1,3-Dimethoxybenzene has been used in the synthesis of novel oxathiane spiroketal donors.[1]
Related compounds
References
- ↑ A. Fascione, Martin; J. Webb, Nicola; A. Kilner, Colin; L. Warriner, Stuart; Bruce Turnbull, W. (2011-12-28). "Stereoselective glycosylations using oxathiane spiroketal glycosyl donors". Merck. 348: 6–13. doi:10.1016/j.carres.2011.07.020. PMID 22200482.
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.