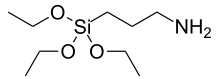
 Structural formula of (3-aminopropyl)triethoxysilane | |
| Names | |
|---|---|
| Preferred IUPAC name
3-(Triethoxysilyl)propan-1-amine | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.011.863 |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C9H23NO3Si | |
| Molar mass | 221.372 g·mol−1 |
| Density | 0.946 g/mL[1] |
| Melting point | −70 °C (−94 °F; 203 K) |
| Boiling point | 217 °C (423 °F; 490 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
(3-Aminopropyl)triethoxysilane (APTES) is an aminosilane frequently used in the process of silanization, the functionalization of surfaces with alkoxysilane molecules. It can also be used for covalent attaching of organic films to metal oxides such as silica and titania.
Use with PDMS
APTES can be used to covalently bond thermoplastics to poly(dimethylsiloxane) (PDMS).[2] Thermoplastics are treated with oxygen plasma to functionalize surface molecules, and subsequently coated with an aqueous 1% by volume APTES solution. PDMS is treated with oxygen plasma and placed in contact with the functionalized thermoplastic surface. A stable, covalent bond forms within 2 minutes.
Silsesquioxane synthesis
Octa(3-aminopropyl)silsesquioxane can be obtained in a one step hydrolytic condensation using APTES and hydrochloric or trifluoromethanesulfonic acid (CF3SO3H).[3]
Use with cell cultures
APTES-functionalized surfaces have been shown to be nontoxic to embryonic rat cardiomyocytes in vitro.[4] Further experimentation is needed to evaluate toxicity to other cell types in extended culture.
Toxicity
APTES is a toxic compound with an MSDS health hazard score of 3. APTES fumes are destructive to the mucous membranes and the upper respiratory tract, and should be used in a fume hood with gloves. If a fume hood is not available, a face shield and full face respirator must be implemented. The target organs of APTES are nerves, liver and kidney.
References
- ↑ "3-Aminopropyltriethoxysilane | 919-30-2". Chemicalbook.com. Retrieved 2012-08-01.
- ↑ Sunkara V., Park D.K., Hwang H., Chantiwas R., Soper S.A., and Cho Y.K. (2011). Simple room temperature bonding of thermoplastics and poly(dimethylsiloxane). Lab on a Chip, 11(5), 962-965
- ↑ Janeta, Mateusz; John, Łukasz; Ejfler, Jolanta; Szafert, Sławomir (2014-11-24). "High-Yield Synthesis of Amido-Functionalized Polyoctahedral Oligomeric Silsesquioxanes by Using Acyl Chlorides". Chemistry: A European Journal. 20 (48): 15966–15974. doi:10.1002/chem.201404153. ISSN 1521-3765. PMID 25302846.
- ↑ Natarajan, Growth and Electrophysiological Properties of Rat Embryonic Cardiomyocytes on Hydroxyl- and Carboxyl-Modified Surfaces, J Biomater Sci Polym Ed 2010