 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
2,3,5,6-Tetrahydroxycyclohexa-2,5-diene-1,4-dione | |
| Other names
Tetroquinone | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.005.706 |
| EC Number |
|
| KEGG | |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C6H4O6 | |
| Molar mass | 172.092 g·mol−1 |
| Appearance | Blue-black crystals |
| Density | 2.609 g/cm3 |
| Boiling point | 370.6 °C (699.1 °F; 643.8 K) at 760 mmHg |
| Slightly soluble in cold water | |
| Hazards | |
| GHS labelling: | |
 | |
| Warning | |
| H315, H319, H335 | |
| P261, P264, P271, P280, P302+P352, P304+P340, P305+P351+P338, P312, P321, P332+P313, P337+P313, P362, P403+P233, P405, P501 | |
| Flash point | 192.1 °C (377.8 °F; 465.2 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
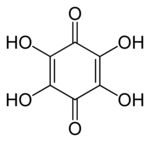
Tetrahydroxy-1,4-benzoquinone, also called tetrahydroxy-p-benzoquinone, tetrahydroxybenzoquinone, or tetrahydroxyquinone (THBQ, THQ), is an organic compound with formula C6O2(OH)4. Its molecular structure consists of a cyclohexadiene ring with four hydroxyl groups and two ketone groups in opposite (para) positions.
The compound gives a light red solution in water,[2] and crystallizes as the glistening bluish-black (but non-conducting) dihydrate C6O2(OH)4·2H2O.[2][3]
The compound can be synthesized from glyoxal[4] or from myo-inositol, a natural compound widely present in plants.[5] THBQ forms an adduct with 4,4′-bipyridine in a 2:3 ratio.[6]
Salts of THBQ
Like most phenols, THBQ is acidic and easily loses the four hydrogen ions from the hydroxyl groups, yielding anions such as C6H2O2−6 and C6O4−6. The latter is symmetric and aromatic, as the double bonds and negative charges are evenly distributed over the six CO groups.
The calcium salt Ca2C6O6 is the dark purple pigment produced from inositol by Chromohalobacter beijerinckii in the fermentation of salt beans, already noted by T. Hof in 1935.[3][7][8][9][10]
The dark purple and insoluble dipotassium salt K2C6H2O6 was prepared by Preisler and Berger in 1942, by oxidizing inositol with nitric acid and reacting the result with potassium carbonate in the presence of oxygen. Reaction of this salt with hydrochloric acid produces THBQ in good yield.[11]
The black tetrapotassium salt K4C6O6 was prepared by West and Niu in 1962, by reacting THBQ with potassium methoxide in methanol. The salt is diamagnetic and the infrared spectrum suggests that the C–C and C–O distances are all equal, with the ring slightly distorted in the "chair" conformation.[12] Partial oxidation of K4C6O6 affords a green, strongly paramagnetic solid, conjectured to be 3K+·C6O3−6, and complete oxidation yields potassium rhodizonate 2K+·C6O2−6.[12]
The greenish-black sodium salt Na4C6O6 was described by Fatiadi and Sanger in 1962.[4]
The dark-violet lithium salt Li4C6O6 has been proposed as an electrode material for batteries as it can be oxidized to the rhodizonate Li2C6O6 and reduced to the hexahydroxybenzene salt Li6C6O6.[5] In the absence of oxygen, Li4C6O6 is stable to about 450 °C and then decomposes leaving a residue of lithium carbonate.[5] Indeed, the rhodizonate appears to disproportionate at about 400 °C into Li4C6O6 and cyclohexanehexone C6O6 that promptly decomposes into carbon monoxide, carbon dioxide, and carbon.[5] Li4C6O6 forms a hydrate Li4C6O6·2H2O that loses its water at about 250 °C.[5]
References
- ↑ "Tetroquinone". The Merck Index (11th ed.). p. 9177.
- 1 2 Klug, H. P. (1965). "The crystal structure of tetrahydroxy-p-benzoquinone". Acta Crystallographica. 19 (6): 983. doi:10.1107/S0365110X65004760.
- 1 2 Miller, M. W. (1961). Microbial Metabolites. McGraw-Hill.
- 1 2 Fatiadi, A. J.; Sanger, W. F. "Tetrahydroxyquinone". Organic Syntheses. 42: 90.; Collective Volume, vol. 5, p. 1011
- 1 2 3 4 5 Chen, H.; Armand, M.; Courty, M.; Jiang, M.; Grey, C. P.; Dolhem, F.; Tarascon, J.-M.; Poizot, P. (2009). "Lithium salt of tetrahydroxybenzoquinone: toward the development of a sustainable Li-ion battery". Journal of the American Chemical Society. 131 (25): 8984–8988. doi:10.1021/ja9024897. PMID 19476355.
- ↑ Cowan, J. A.; Howard, J. A. K.; Leech, M. A. (2001). "Interpenetrating supramolecular lattices in 4,4′-bipyridine-2,3,5,6-tetrahydroxy-1,4-benzoquinone (3/2)". Acta Crystallographica Section C. 57 (10): 1196–1198. doi:10.1107/S0108270101011647. PMID 11600783.
- ↑ Hof, T. (1935). Recueil des travaux botaniques néerlandais [Collection of Dutch botanical works] (in French). Vol. 32. New Zealand: F.E. Macdonald; Dutch Botanical Society. p. 92. OCLC 312975183.
{{cite book}}: CS1 maint: date and year (link) - ↑ Kluyver, A. J.; Hof, T.; Boezaardt, A. G. J. (1939). "On the pigment of Pseudomonas beijerinckii Hof (Chromohalobacter beijerinckii)". Enzymologia. 7 (257).
{{cite journal}}: CS1 maint: date and year (link) - ↑ Elvehjem, C. A. (June 1947). "Vitamins and Hormones—Advances in Research and Applications". American Journal of Public Health and the Nation's Health. 37 (6): 776. doi:10.2105/AJPH.37.6.776. ISSN 0002-9572. PMC 1623712.
- ↑ Underkofler, L. A.; Hickey, R. J. (1954). Industrial Fermentations.
- ↑ Preisler, P. W.; Berger, L. (1942). "Preparation of tetrahydroxyquinone and rhodizonic acid salts from the product of the oxidation of inositol with nitric acid". Journal of the American Chemical Society. 64 (1): 67–69. doi:10.1021/ja01253a016.
- 1 2 West, R.; Niu, H. Y. (1962). "Symmetrical Resonance Stabilized Anions, CnO−m
n. II. K4C6O6 and Evidence for C6O−3
6". Journal of the American Chemical Society. 84 (7): 1324–1325. doi:10.1021/ja00866a068.