.jpg.webp)

| Part of a series on |
| Water salinity |
|---|
 |
| Salinity levels |
|
Fresh water (< 0.05%) Brackish water (0.05–3%) Saline water (3–5%) Brine (> 5% up to 26%–28% max) |
| Bodies of water |
Seawater, or sea water, is water from a sea or ocean. On average, seawater in the world's oceans has a salinity of about 3.5% (35 g/L, 35 ppt, 600 mM). This means that every kilogram (roughly one liter by volume) of seawater has approximately 35 grams (1.2 oz) of dissolved salts (predominantly sodium (Na+
) and chloride (Cl−
) ions). The average density at the surface is 1.025 kg/L. Seawater is denser than both fresh water and pure water (density 1.0 kg/L at 4 °C (39 °F)) because the dissolved salts increase the mass by a larger proportion than the volume. The freezing point of seawater decreases as salt concentration increases. At typical salinity, it freezes at about −2 °C (28 °F).[1] The coldest seawater still in the liquid state ever recorded was found in 2010, in a stream under an Antarctic glacier: the measured temperature was −2.6 °C (27.3 °F).[2]
Seawater pH is typically limited to a range between 7.5 and 8.4.[3] However, there is no universally accepted reference pH-scale for seawater and the difference between measurements based on different reference scales may be up to 0.14 units.[4]
Properties
Salinity

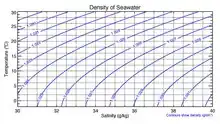
Although the vast majority of seawater has a salinity of between 31 and 38 g/kg, that is 3.1–3.8%, seawater is not uniformly saline throughout the world. Where mixing occurs with freshwater runoff from river mouths, near melting glaciers or vast amounts of precipitation (e.g. monsoon), seawater can be substantially less saline. The most saline open sea is the Red Sea, where high rates of evaporation, low precipitation and low river run-off, and confined circulation result in unusually salty water. The salinity in isolated bodies of water can be considerably greater still – about ten times higher in the case of the Dead Sea. Historically, several salinity scales were used to approximate the absolute salinity of seawater. A popular scale was the "Practical Salinity Scale" where salinity was measured in "practical salinity units (PSU)". The current standard for salinity is the "Reference Salinity" scale [6] with the salinity expressed in units of "g/kg".
Density
The density of surface seawater ranges from about 1020 to 1029 kg/m3, depending on the temperature and salinity. At a temperature of 25 °C, the salinity of 35 g/kg and 1 atm pressure, the density of seawater is 1023.6 kg/m3.[7][8] Deep in the ocean, under high pressure, seawater can reach a density of 1050 kg/m3 or higher. The density of seawater also changes with salinity. Brines generated by seawater desalination plants can have salinities up to 120 g/kg. The density of typical seawater brine of 120 g/kg salinity at 25 °C and atmospheric pressure is 1088 kg/m3.[7][8]
pH value
The pH value at the surface of oceans in pre-industrial time (before 1850) was around 8.2.[9] Since then, it has been decreasing due to a human-caused process called ocean acidification that is related to carbon dioxide emissions: Between 1950 and 2020, the average pH of the ocean surface fell from approximately 8.15 to 8.05.[10]
The pH value of seawater is naturally as low as 7.8 in deep ocean waters as a result of degradation of organic matter in these waters.[11] It can be as high as 8.4 in surface waters in areas of high biological productivity.[12]
Measurement of pH is complicated by the chemical properties of seawater, and several distinct pH scales exist in chemical oceanography.[13] There is no universally accepted reference pH-scale for seawater and the difference between measurements based on different reference scales may be up to 0.14 units.[4]
Chemical composition
Seawater contains more dissolved ions than all types of freshwater.[14] However, the ratios of solutes differ dramatically. For instance, although seawater contains about 2.8 times more bicarbonate than river water, the percentage of bicarbonate in seawater as a ratio of all dissolved ions is far lower than in river water. Bicarbonate ions constitute 48% of river water solutes but only 0.14% for seawater.[14][15] Differences like these are due to the varying residence times of seawater solutes; sodium and chloride have very long residence times, while calcium (vital for carbonate formation) tends to precipitate much more quickly.[15] The most abundant dissolved ions in seawater are sodium, chloride, magnesium, sulfate and calcium.[16] Its osmolarity is about 1000 mOsm/L.[17]
Small amounts of other substances are found, including amino acids at concentrations of up to 2 micrograms of nitrogen atoms per liter,[18] which are thought to have played a key role in the origin of life.
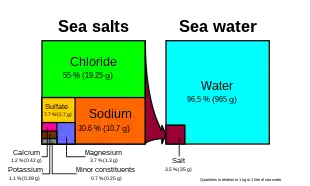
55%, Na+
30.6%, SO2−
4 7.7%, Mg2+
3.7%, Ca2+
1.2%, K+
1.1%, Other 0.7%. Note that the diagram is only correct when in units of wt/wt, not wt/vol or vol/vol.
| Element | Percent by mass |
|---|---|
| Oxygen | 85.84 |
| Hydrogen | 10.82 |
| Chlorine | 1.94 |
| Sodium | 1.08 |
| Magnesium | 0.1292 |
| Sulfur | 0.091 |
| Calcium | 0.04 |
| Potassium | 0.04 |
| Bromine | 0.0067 |
| Carbon | 0.0028 |
| Component | Concentration (mol/kg) |
|---|---|
| H 2O | 53.6 |
| Cl− | 0.546 |
| Na+ | 0.469 |
| Mg2+ | 0.0528 |
| SO2− 4 | 0.0282 |
| Ca2+ | 0.0103 |
| K+ | 0.0102 |
| CT | 0.00206 |
| Br− | 0.000844 |
| BT | 0.000416 |
| Sr2+ | 0.000091 |
| F− | 0.000068 |
Microbial components
Research in 1957 by the Scripps Institution of Oceanography sampled water in both pelagic and neritic locations in the Pacific Ocean. Direct microscopic counts and cultures were used, the direct counts in some cases showing up to 10 000 times that obtained from cultures. These differences were attributed to the occurrence of bacteria in aggregates, selective effects of the culture media, and the presence of inactive cells. A marked reduction in bacterial culture numbers was noted below the thermocline, but not by direct microscopic observation. Large numbers of spirilli-like forms were seen by microscope but not under cultivation. The disparity in numbers obtained by the two methods is well known in this and other fields.[20] In the 1990s, improved techniques of detection and identification of microbes by probing just small snippets of DNA, enabled researchers taking part in the Census of Marine Life to identify thousands of previously unknown microbes usually present only in small numbers. This revealed a far greater diversity than previously suspected, so that a litre of seawater may hold more than 20,000 species. Mitchell Sogin from the Marine Biological Laboratory feels that "the number of different kinds of bacteria in the oceans could eclipse five to 10 million."[21]
Bacteria are found at all depths in the water column, as well as in the sediments, some being aerobic, others anaerobic. Most are free-swimming, but some exist as symbionts within other organisms – examples of these being bioluminescent bacteria. Cyanobacteria played an important role in the evolution of ocean processes, enabling the development of stromatolites and oxygen in the atmosphere.
Some bacteria interact with diatoms, and form a critical link in the cycling of silicon in the ocean. One anaerobic species, Thiomargarita namibiensis, plays an important part in the breakdown of hydrogen sulfide eruptions from diatomaceous sediments off the Namibian coast, and generated by high rates of phytoplankton growth in the Benguela Current upwelling zone, eventually falling to the seafloor.
Bacteria-like Archaea surprised marine microbiologists by their survival and thriving in extreme environments, such as the hydrothermal vents on the ocean floor. Alkalotolerant marine bacteria such as Pseudomonas and Vibrio spp. survive in a pH range of 7.3 to 10.6, while some species will grow only at pH 10 to 10.6.[22] Archaea also exist in pelagic waters and may constitute as much as half the ocean's biomass, clearly playing an important part in oceanic processes.[23] In 2000 sediments from the ocean floor revealed a species of Archaea that breaks down methane, an important greenhouse gas and a major contributor to atmospheric warming.[24] Some bacteria break down the rocks of the sea floor, influencing seawater chemistry. Oil spills, and runoff containing human sewage and chemical pollutants have a marked effect on microbial life in the vicinity, as well as harbouring pathogens and toxins affecting all forms of marine life. The protist dinoflagellates may at certain times undergo population explosions called blooms or red tides, often after human-caused pollution. The process may produce metabolites known as biotoxins, which move along the ocean food chain, tainting higher-order animal consumers.
Pandoravirus salinus, a species of very large virus, with a genome much larger than that of any other virus species, was discovered in 2013. Like the other very large viruses Mimivirus and Megavirus, Pandoravirus infects amoebas, but its genome, containing 1.9 to 2.5 megabases of DNA, is twice as large as that of Megavirus, and it differs greatly from the other large viruses in appearance and in genome structure.
In 2013 researchers from Aberdeen University announced that they were starting a hunt for undiscovered chemicals in organisms that have evolved in deep sea trenches, hoping to find "the next generation" of antibiotics, anticipating an "antibiotic apocalypse" with a dearth of new infection-fighting drugs. The EU-funded research will start in the Atacama Trench and then move on to search trenches off New Zealand and Antarctica.[25]
The ocean has a long history of human waste disposal on the assumption that its vast size makes it capable of absorbing and diluting all noxious material.[26] While this may be true on a small scale, the large amounts of sewage routinely dumped has damaged many coastal ecosystems, and rendered them life-threatening. Pathogenic viruses and bacteria occur in such waters, such as Escherichia coli, Vibrio cholerae the cause of cholera, hepatitis A, hepatitis E and polio, along with protozoans causing giardiasis and cryptosporidiosis. These pathogens are routinely present in the ballast water of large vessels, and are widely spread when the ballast is discharged.[27]
Other parameters
The speed of sound in seawater is about 1,500 m/s (whereas the speed of sound is usually around 330 m/s in air at roughly 101.3 kPa pressure, 1 atmosphere), and varies with water temperature, salinity, and pressure. The thermal conductivity of seawater is 0.6 W/mK at 25 °C and a salinity of 35 g/kg.[28] The thermal conductivity decreases with increasing salinity and increases with increasing temperature.[29]
Origin and history
The water in the sea was thought to come from the Earth's volcanoes, starting 4 billion years ago, released by degassing from molten rock.[30]: 24–25 More recent work suggests much of the Earth's water may come from comets.[31]
Scientific theories behind the origins of sea salt started with Sir Edmond Halley in 1715, who proposed that salt and other minerals were carried into the sea by rivers after rainfall washed it out of the ground. Upon reaching the ocean, these salts concentrated as more salt arrived over time (see Hydrologic cycle). Halley noted that most lakes that don't have ocean outlets (such as the Dead Sea and the Caspian Sea, see endorheic basin), have high salt content. Halley termed this process "continental weathering".
Halley's theory was partly correct. In addition, sodium leached out of the ocean floor when the ocean formed. The presence of salt's other dominant ion, chloride, results from outgassing of chloride (as hydrochloric acid) with other gases from Earth's interior via volcanos and hydrothermal vents. The sodium and chloride ions subsequently became the most abundant constituents of sea salt.
Ocean salinity has been stable for billions of years, most likely as a consequence of a chemical/tectonic system which removes as much salt as is deposited; for instance, sodium and chloride sinks include evaporite deposits, pore-water burial, and reactions with seafloor basalts.[15]: 133
Human impacts
Climate change, rising levels of carbon dioxide in Earth's atmosphere, excess nutrients, and pollution in many forms are altering global oceanic geochemistry. Rates of change for some aspects greatly exceed those in the historical and recent geological record. Major trends include an increasing acidity, reduced subsurface oxygen in both near-shore and pelagic waters, rising coastal nitrogen levels, and widespread increases in mercury and persistent organic pollutants. Most of these perturbations are tied either directly or indirectly to human fossil fuel combustion, fertilizer, and industrial activity. Concentrations are projected to grow in coming decades, with negative impacts on ocean biota and other marine resources.[32]
One of the most striking features of this is ocean acidification, resulting from increased CO2 uptake of the oceans related to higher atmospheric concentration of CO2 and higher temperatures,[33] because it severely affects coral reefs, mollusks, echinoderms and crustaceans (see coral bleaching).
Human consumption
Accidentally consuming small quantities of clean seawater is not harmful, especially if the seawater is taken along with a larger quantity of fresh water. However, drinking seawater to maintain hydration is counterproductive; more water must be excreted to eliminate the salt (via urine) than the amount of water obtained from the seawater itself.[34] In normal circumstances, it would be considered ill-advised to consume large amounts of unfiltered seawater.
The renal system actively regulates the levels of sodium and chloride in the blood within a very narrow range around 9 g/L (0.9% by mass).
In most open waters concentrations vary somewhat around typical values of about 3.5%, far higher than the body can tolerate and most beyond what the kidney can process. A point frequently overlooked in claims that the kidney can excrete NaCl in Baltic concentrations of 2% (in arguments to the contrary) is that the gut cannot absorb water at such concentrations, so that there is no benefit in drinking such water. The salinity of Baltic surface water, however, is never 2%. It is 0.9% or less, and thus never higher than that of bodily fluids. Drinking seawater temporarily increases blood's NaCl concentration. This signals the kidney to excrete sodium, but seawater's sodium concentration is above the kidney's maximum concentrating ability. Eventually the blood's sodium concentration rises to toxic levels, removing water from cells and interfering with nerve conduction, ultimately producing fatal seizure and cardiac arrhythmia.
Survival manuals consistently advise against drinking seawater.[35] A summary of 163 life raft voyages estimated the risk of death at 39% for those who drank seawater, compared to 3% for those who did not. The effect of seawater intake on rats confirmed the negative effects of drinking seawater when dehydrated.[36]
The temptation to drink seawater was greatest for sailors who had expended their supply of fresh water and were unable to capture enough rainwater for drinking. This frustration was described famously by a line from Samuel Taylor Coleridge's The Rime of the Ancient Mariner:
Water, water, everywhere,
And all the boards did shrink;
Water, water, everywhere,
Nor any drop to drink.
Although humans cannot survive on seawater, some people claim that up to two cups a day, mixed with fresh water in a 2:3 ratio, produces no ill effect. The French physician Alain Bombard survived an ocean crossing in a small Zodiak rubber boat using mainly raw fish meat, which contains about 40% water (like most living tissues), as well as small amounts of seawater and other provisions harvested from the ocean. His findings were challenged, but an alternative explanation was not given. In his 1948 book The Kon-Tiki Expedition, Thor Heyerdahl reported drinking seawater mixed with fresh in a 2:3 ratio during the 1947 expedition.[37] A few years later, another adventurer, William Willis, claimed to have drunk two cups of seawater and one cup of fresh per day for 70 days without ill effect when he lost part of his water supply.[38]
During the 18th century, Richard Russell advocated the medical use of this practice in the UK,[39] and René Quinton expanded the advocation of this practice to other countries, notably France, in the 20th century. Currently, it is widely practiced in Nicaragua and other countries, supposedly taking advantage of the latest medical discoveries.[40][41]
Most oceangoing vessels desalinate potable water from seawater using processes such as vacuum distillation or multi-stage flash distillation in an evaporator, or, more recently, reverse osmosis. These energy-intensive processes were not usually available during the Age of Sail. Larger sailing warships with large crews, such as Nelson's HMS Victory, were fitted with distilling apparatus in their galleys.[42] Animals such as fish, whales, sea turtles, and seabirds, such as penguins and albatrosses, have adapted to living in a high-saline habitat. For example, sea turtles and saltwater crocodiles remove excess salt from their bodies through their tear ducts.[43]
Mineral extraction
Minerals have been extracted from seawater since ancient times. Currently the four most concentrated metals – Na, Mg, Ca and K – are commercially extracted from seawater.[44] During 2015 in the US 63% of magnesium production came from seawater and brines.[45] Bromine is also produced from seawater in China and Japan.[46] Lithium extraction from seawater was tried in the 1970s, but the tests were soon abandoned. The idea of extracting uranium from seawater has been considered at least from the 1960s, but only a few grams of uranium were extracted in Japan in the late 1990s.[47] The main issue is not one of technological feasibility but that current prices on the uranium market for uranium from other sources are about three to five times lower than the lowest price achieved by seawater extraction.[48][49] Similar issues hamper the use of reprocessed uranium and are often brought forth against nuclear reprocessing and the manufacturing of MOX fuel as economically unviable.
Standard
ASTM International has an international standard for artificial seawater: ASTM D1141-98 (Original Standard ASTM D1141-52). It is used in many research testing labs as a reproducible solution for seawater such as tests on corrosion, oil contamination, and detergency evaluation.[50]
See also
- Brine – Concentrated solution of salt in water
- Brine mining – Extracting materials from saltwater
- Brackish water – Water with salinity between freshwater and seawater
- Fresh water – Naturally occurring water with low amounts of dissolved salts
- Ocean color – Explanation of the color of oceans and ocean color remote sensing
- Saline water – Water that contains a high concentration of dissolved salts
- Sea ice – Outcome of seawater as it freezes
- Seawater pH – Measure of the level of acidity or basicity of an aqueous solution
- Surface tension of seawater – Tendency of a liquid surface to shrink to reduce surface area
- Thalassotherapy
- Thermohaline circulation – Part of large-scale ocean circulation
- CORA dataset – Oceanographic temperature and salinity dataset global ocean salinity
References
- ↑ "U.S. Office of Naval Research Ocean, Water: Temperature". Archived from the original on 12 December 2007.
- ↑ Sylte, Gudrun Urd (24 May 2010). "Den aller kaldaste havstraumen". forskning.no (in Norwegian). Archived from the original on 6 March 2012. Retrieved 24 May 2010.
- ↑ Chester, Jickells, Roy, Tim (2012). Marine Geochemistry. Blackwell Publishing. ISBN 978-1-118-34907-6.
{{cite book}}: CS1 maint: multiple names: authors list (link) - 1 2 Stumm, W, Morgan, J. J. (1981) Aquatic Chemistry, An Introduction Emphasizing Chemical Equilibria in Natural Waters. John Wiley & Sons. pp. 414–416. ISBN 0471048313.
- ↑ "World Ocean Atlas 2009". NOAA. Retrieved 5 December 2012.
- ↑ Millero, Frank J.; Feistel, Rainer; Wright, Daniel G.; McDougall, Trevor J. (January 2008). "The composition of Standard Seawater and the definition of the Reference-Composition Salinity Scale". Deep Sea Research Part I: Oceanographic Research Papers. 55 (1): 50–72. Bibcode:2008DSRI...55...50M. doi:10.1016/j.dsr.2007.10.001.
- 1 2 Nayar, Kishor G.; Sharqawy, Mostafa H.; Banchik, Leonardo D.; Lienhard V, John H. (July 2016). "Thermophysical properties of seawater: A review and new correlations that include pressure dependence". Desalination. 390: 1–24. doi:10.1016/j.desal.2016.02.024.
- 1 2 "Thermophysical properties of seawater". Department of Mechanical Engineering, Massachusetts Institute of Technology. Retrieved 24 February 2017.
- ↑ Arias, P.A., N. Bellouin, E. Coppola, R.G. Jones, G. Krinner, J. Marotzke, V. Naik, M.D. Palmer, G.-K. Plattner, J. Rogelj, M. Rojas, J. Sillmann, T. Storelvmo, P.W. Thorne, B. Trewin, K. Achuta Rao, B. Adhikary, R.P. Allan, K. Armour, G. Bala, R. Barimalala, S. Berger, J.G. Canadell, C. Cassou, A. Cherchi, W. Collins, W.D. Collins, S.L. Connors, S. Corti, F. Cruz, F.J. Dentener, C. Dereczynski, A. Di Luca, A. Diongue Niang, F.J. Doblas-Reyes, A. Dosio, H. Douville, F. Engelbrecht, V. Eyring, E. Fischer, P. Forster, B. Fox-Kemper, J.S. Fuglestvedt, J.C. Fyfe, et al., 2021: Technical Summary Archived 21 July 2022 at the Wayback Machine. In Climate Change 2021: The Physical Science Basis. Contribution of Working Group I to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change Archived 9 August 2021 at the Wayback Machine [Masson-Delmotte, V., P. Zhai, A. Pirani, S.L. Connors, C. Péan, S. Berger, N. Caud, Y. Chen, L. Goldfarb, M.I. Gomis, M. Huang, K. Leitzell, E. Lonnoy, J.B.R. Matthews, T.K. Maycock, T. Waterfield, O. Yelekçi, R. Yu, and B. Zhou (eds.)]. Cambridge University Press, Cambridge, United Kingdom and New York, NY, USA, pp. 33−144.
- ↑ Terhaar, Jens; Frölicher, Thomas L.; Joos, Fortunat (2023). "Ocean acidification in emission-driven temperature stabilization scenarios: the role of TCRE and non-CO2 greenhouse gases". Environmental Research Letters. 18 (2): 024033. Bibcode:2023ERL....18b4033T. doi:10.1088/1748-9326/acaf91. ISSN 1748-9326. S2CID 255431338.
- ↑ Emerson, Steven; Hedges, John (24 April 2008). "Chapter 4: Carbonate chemistry". Chemical Oceanography and the Marine Carbon Cycle (1 ed.). Cambridge University Press. doi:10.1017/cbo9780511793202. ISBN 978-0-521-83313-4.
- ↑ Chester, R.; Jickells, Tim (2012). "Chapter 9: Nutrients, oxygen, organic carbon and the carbon cycle in seawater". Marine geochemistry (3rd ed.). Chichester, West Sussex, UK: Wiley/Blackwell. ISBN 978-1-118-34909-0. OCLC 781078031.
- ↑ Zeebe, R. E. and Wolf-Gladrow, D. (2001) CO2 in seawater: equilibrium, kinetics, isotopes, Elsevier Science B.V., Amsterdam, Netherlands ISBN 0-444-50946-1
- 1 2 Gale, Thomson. "Ocean Chemical Processes". Retrieved 2 December 2006.
- 1 2 3 Pinet, Paul R. (1996). Invitation to Oceanography. St. Paul: West Publishing Company. pp. 126, 134–135. ISBN 978-0-314-06339-7.
- ↑ Hogan, C. Michael (2010). "Calcium", eds. A. Jorgensen, C. Cleveland. Encyclopedia of Earth. Some evidence shows the potential for fairly regular ratios of elements maintained across surface oceans in a phenomenon known as the Redfield Ratio. National Council for Science and the Environment.
- ↑ "Osmolarity of sea water".
- ↑ Tada, K.; Tada, M.; Maita, Y. (1998). "Dissolved free amino acids in coastal seawater using a modified fluorometric method" (PDF). Journal of Oceanography. 54 (4): 313–321. doi:10.1007/BF02742615. S2CID 26231863. Archived from the original (PDF) on 21 January 2021. Retrieved 28 August 2015.
- ↑ DOE (1994). "5" (PDF). In A. G. Dickson; C. Goyet (eds.). Handbook of methods for the analysis of the various parameters of the carbon dioxide system in sea water. 2. ORNL/CDIAC-74. Archived from the original (PDF) on 25 May 2011. Retrieved 18 May 2006.
- ↑ Jannasch, Holger W.; Jones, Galen E. (1959). "Bacterial Populations in Sea Water as Determined by Different Methods of Enumeration". Limnology and Oceanography. 4 (2): 128–139. Bibcode:1959LimOc...4..128J. doi:10.4319/lo.1959.4.2.0128.
- ↑ "Ocean Microbe Census Discovers Diverse World of Rare Bacteria". ScienceDaily. 2 September 2006. Retrieved 13 May 2013.
- ↑ Maeda, M.; Taga, N. (31 March 1980). "Alkalotolerant and Alkalophilic Bacteria in Seawater". Marine Ecology Progress Series. 2: 105–108. Bibcode:1980MEPS....2..105M. doi:10.3354/meps002105.
- ↑ Cheung, Louisa (31 July 2006). "Thousands of microbes in one gulp". BBC News. Retrieved 13 May 2013.
- ↑ Leslie, Mitchell (5 October 2000). "The Case of the Missing Methane". ScienceNOW. American Association for the Advancement of Science. Archived from the original on 26 May 2013. Retrieved 13 May 2013.
- ↑ "Antibiotics search to focus on sea bed". BBC News. 14 February 2013. Retrieved 13 May 2013.
- ↑ Panel On Radioactivity In The Marine Environment, National Research Council (U.S.) (1971). Radioactivity in the marine environment. National Academies. p. 36. ISBN 9780309018654.
{{cite book}}:|work=ignored (help) - ↑ Hoyle, Brian D.; Robinson, Richard. "Microbes in the Ocean". Water Encyclopedia.
- ↑ Sharqawy, Mostafa H.; Lienhard V, John H.; Zubair, Syed M. (April 2010). "The thermophysical properties of seawater: A review of existing correlations and data" (PDF). Desalination and Water Treatment. 16 (1–3): 354–380. doi:10.5004/dwt.2010.1079. hdl:1721.1/69157.
- ↑ "Thermal conductivity of seawater and its concentrates". Retrieved 17 October 2010.
- ↑ Stow, Dorrik (2004). Encyclopedia of the Oceans. Oxford University Press. ISBN 978-0-19-860687-1.
- ↑ Cowen, Ron (5 October 2011). "Comets take pole position as water bearers". Nature. doi:10.1038/news.2011.579. Retrieved 10 September 2013.
- ↑ Doney, Scott C. (18 June 2010). "The Growing Human Footprint on Coastal and Open-Ocean Biogeochemistry". Science. 328 (5985): 1512–1516. Bibcode:2010Sci...328.1512D. doi:10.1126/science.1185198. PMID 20558706. S2CID 8792396.
- ↑ Doney, Scott C.; Fabry, Victoria J.; Feely, Richard A.; Kleypas, Joan A. (1 January 2009). "Ocean Acidification: The Other CO2 Problem". Annual Review of Marine Science. 1 (1): 169–192. Bibcode:2009ARMS....1..169D. doi:10.1146/annurev.marine.010908.163834. PMID 21141034. S2CID 402398.
- ↑ "Can humans drink seawater?". National Ocean Service (NOAA). 26 February 2021.
- ↑ "29" (PDF). Shipboard Medicine. Archived from the original (PDF) on 22 June 2007. Retrieved 17 October 2010.
- ↑ Etzion, Z.; Yagil, R. (1987). "Metabolic effects in rats drinking increasing concentrations of seawater". Comp Biochem Physiol A. 86 (1): 49–55. doi:10.1016/0300-9629(87)90275-1. PMID 2881655.
- ↑ Heyerdahl, Thor; Lyon, F. H. (translator) (1950). Kon-Tiki: Across the Pacific by Raft. Rand McNally & Company, Chicago, Ill.
- ↑ King, Dean (2004). Skeletons on the Zahara: a true story of survival. New York: Back Bay Books. p. 74. ISBN 978-0-316-15935-7.
- ↑ "History of the medical use of sea water in U.K. in 18th century".
- ↑ Martin, Francisco (2020). "chapter 12: Medical use of sea water in Nicaragua". Drinking Sea Water. ISBN 979-8666741658.
- ↑ "Medical use of sea water in Nicaragua".
- ↑ Rippon, P. M., Commander, RN (1998). The evolution of engineering in the Royal Navy. Vol. 1: 1827–1939. Spellmount. pp. 78–79. ISBN 978-0-946771-55-4.
{{cite book}}: CS1 maint: multiple names: authors list (link) - ↑ Dennis, Jerry (23 September 2014). The Bird in the Waterfall: Exploring the Wonders of Water. Diversion Books. ISBN 9781940941547.
- ↑ Loganathan, Paripurnanda; Naidu, Gayathri; Vigneswaran, Saravanamuthu (2017). "Mining valuable minerals from seawater: a critical review". Environmental Science: Water Research & Technology. 3 (1): 37–53. doi:10.1039/C6EW00268D. hdl:10453/121701.
- ↑ Campbell, Keith. "Over 40 minerals and metals contained in seawater, their extraction likely to increase in the future". Mining Weekly. Retrieved 8 February 2023.
- ↑ Global Bromine Industry And Its Outlook
- ↑ Ugo Bardi (2008). "Mining the Oceans: Can We Extract Minerals from Seawater?". theoildrum.com. Retrieved 8 February 2023.
- ↑ "Viability of Uranium Extraction from Sea Water".
- ↑ "Cost-effective method of extracting uranium from seawater promises limitless nuclear power". 14 June 2018.
- ↑ "ASTM D1141-98(2013)". ASTM. Retrieved 17 August 2013.
External links
Tables
- Tables and software for thermophysical properties of seawater, MIT
- G. W. C Kaye, T. H. Laby (1995). "Physical properties of sea water". Tables of physical and chemical constants (16th ed.). Archived from the original on 8 May 2019.