| Hypersensitivity Pneumonitis | |
|---|---|
| Other names | Allergic alveolitis, bagpipe lung, extrinsic allergic alveolitis (EAA) |
 | |
| High magnification photomicrograph of a lung biopsy taken showing chronic hypersensitivity pneumonitis (H&E), showing mild thickening of the walls of the small air sacs by invasion of white blood cells. A multinucleated giant cell, seen within the walls of the air sacs to the right of the picture halfway down, is an important clue to the correct diagnosis. | |
| Specialty | Pulmonology, Occupational Safety and Health, Occupational Medicine, Immunology |
| Symptoms | Dyspnea, fever, chills, cough, fatigue, weight loss |
| Complications | Chronic interstitial lung disease |
| Usual onset | Hours to years depending on type of exposure. |
| Duration | Hours to years depending on type of exposure. |
| Types | Acute, Subacute, Chronic or Fibrotic vs. Non-fibrotic |
| Causes | Variety of respirable antigens found in farming, food processing, handing of animals, and different occupational settings |
| Diagnostic method | Based on home and environmental exposure history taking followed by imaging, spirometry, serology, bronchoscopy, and even lung biopsy. |
| Differential diagnosis | Idiopathic Pulmonary Fibrosis, Occupational asthma, Sarcoidosis |
| Prevention | Avoidance of antigen exposure |
| Treatment | Avoidance of antigen exposure and sometimes steroids |
Hypersensitivity pneumonitis (HP) or extrinsic allergic alveolitis (EAA) is a syndrome caused by the repetitive inhalation of antigens from the environment in susceptible or sensitized people.[1][2] Common antigens include molds, bacteria, bird droppings, bird feathers, agricultural dusts, bioaerosols and chemicals from paints or plastics.[3] People affected by this type of lung inflammation (pneumonitis) are commonly exposed to the antigens by their occupations, hobbies, the environment and animals.[4][3] The inhaled antigens produce a hypersensitivity immune reaction causing inflammation of the airspaces (alveoli) and small airways (bronchioles) within the lung.[4] Hypersensitivity pneumonitis may eventually lead to interstitial lung disease.[5]
Signs and symptoms
Hypersensitivity pneumonitis (HP) can be categorized as acute, subacute, and chronic based on the duration of the illness.[6][3]
Acute
In the acute form of HP dose of antigen exposure tends to be very high but only for a short duration.[2] Symptoms may develop 4–6 hours following heavy exposure to the provoking antigen. Symptoms include fever, chills, malaise, cough, chest tightness, dyspnea, rash, swelling and headache. Symptoms resolve within 12 hours to several days upon cessation of exposure.[1]
Subacute
Patients with subacute HP gradually develop a productive cough, dyspnea, fatigue, anorexia, weight loss, and pleurisy. Symptoms are similar to the acute form of the disease, but are less severe and last longer. Findings may be present in patients who have experienced repeated acute attacks.
Chronic
In chronic HP, dose of the antigen tends to be low volume but for a longer duration.[2] Patients often lack a history of acute episodes. They have an insidious onset of cough, progressive dyspnea, fatigue, and weight loss. This is associated with partial to complete but gradual reversibility. Avoiding any further exposure is recommended. Clubbing is observed in 50% of patients. Tachypnea, respiratory distress, and inspiratory crackles over lower lung fields often are present.[1] In fact, hypersensitivity pneumonitis can often resemble IPF in terms of pulmonary fibrosis in that many patients experience hypoxemia.[3]
Epidemiology
Although the prevalence of hypersensitivity pneumonitis is not established it is thought to be low.[7] Data collection limitations are a result of difficulty in diagnosis, sub-clinical presentations that go undetected and variability in climate, region and proximity to local industries.[7] The most common types are bird fancier's and farmer's lung.[8][7] Interestingly, cigarette smoking appears to be protective against the disease.[3]
Pathophysiology
Hypersensitivity pneumonitis is caused by an exaggerated immune response (hypersensitivity). Type III hypersensitivity and type IV hypersensitivity can both occur depending on the cause.[9][10] In general, acute HP is suspected to be attributed to a type III hypersensitivity while the subacute and chronic types are suspected to be caused by T cell infiltration and granuloma formation.[11] Because different people react variably to antigen exposure, the exact mechanism is unclear but genetic and host factors are likely at play.[2] The two hit hypothesis is often toted in the literature to explain why some people have a normal reaction to an antigenic exposure without clinical findings while others experience an exaggerated immune response. The "first hit" in the hypothesis is proposed to be genetic susceptibility and surrounding environmental factors and the "second hit" would be the introduction of the antigen into the respiratory system which causes the exaggerated immune response.[8]
Diagnosis
The diagnosis is made through clinical judgement using a combination of findings because there does not exist a single, universal diagnostic criteria for the disease.[12][3] The diagnosis is most commonly ascertained first with a detailed exposure history followed by a battery of clinical tests including: imaging, histopathology, pulmonary function testing, serology, bronchoscopy, and more.[3][12] In 2020, official guidelines were published by American Thoracic Society, Japanese Respiratory Society, and Asociación Latinoamericana del Tórax which provides a systematic approach to the diagnosis of HP that relies on high-resolution computed tomography.[12]
Exposure History
A detailed occupational, home and environmental exposure history is the first step in diagnosis. Unfortunately, only 60% of inciting antigens are identified in exposure assessment.[12] Re-exposure to the antigen can help aid in diagnosis.[3] Standardized questionnaires have been created to help in obtaining an exposure history although no official questionnaire has been purported.[13] It has been recommended that the questionnaire administered should be relevant to the region in which the exposure has potentially occurred.[14]
Detailed exposure assessments are warranted in the cause of damp indoor environments which have the potential to propagate mold throughout the dwelling. The decision to enlist an industrial hygienist should be made based on the answers to evidence based questions during the environmental assessment.[14] The industrial hygienist or environmental scientist will make the decision to conduct additional sampling.[14]
Types
Hypersensitivity pneumonitis may also be called many different names, based on the provoking antigen. These include:
| Type[15] | Specific antigen | Exposure |
|---|---|---|
| Bird fancier's lung Also called bird breeder's lung, pigeon breeder's lung, and poultry worker's lung |
Avian proteins | Feathers and bird droppings [16] |
| Bagassosis Exposure to moldy molasses |
Thermophilic actinomycetes[16] | Moldy bagasse (pressed sugarcane) |
| Cephalosporium HP | Cephalosporium | Contaminated basements (from sewage) |
| Cheese-washer's lung | Penicillium casei[16] or P. roqueforti | Cheese casings |
| Chemical worker's lung – Isocyanate HP | Toluene diisocyanate (TDI), Hexamethylene diisocyanate (HDI), or Methylene bisphenyl isocyanate (MDI) | Paints, resins, and polyurethane foams |
| Chemical worker's lung[16] – Trimellitic anhydride (TMA) HP | Trimellitic anhydride[16] | Plastics, resins, and paints |
| Coffee worker's lung | Coffee bean protein | Coffee bean dust |
| Compost lung | Aspergillus | Compost |
| Detergent worker's disease | Bacillus subtilis enzymes | Detergent |
| Farmer's lung | The molds
The bacteria
|
Moldy hay |
| Hot tub lung | Mycobacterium avium complex | Mist from hot tubs |
| Humidifier lung | The bacteria
The fungi The amoebae
|
Mist generated by a machine from standing water |
| Japanese summer house HP Also called Japanese summer-type HP | Trichosporon cutaneum | Damp wood and mats |
| Laboratory worker's lung | Male rat urine protein | Laboratory rats |
| Lycoperdonosis | Puffball spores | Spore dust from mature puffballs[17] |
| Malt worker's lung | Aspergillus clavatus[16] | Moldy barley |
| Maple bark disease | Cryptostroma corticale[16] | Moldy maple bark |
| Metalworking fluids HP | Nontuberculous mycobacteria | Mist from metalworking fluids |
| Miller's lung | Sitophilus granarius (wheat weevil)[16] | Dust-contaminated grain[16] |
| Mollusc shell HP | Aquatic animal proteins | Mollusc shell dust |
| Mushroom worker's lung | Thermophilic actinomycetes | Mushroom compost |
| Peat moss worker's lung | Caused by Monocillium sp. and Penicillium citreonigrum | Peat moss |
| Pituitary snuff taker's lung | Pituitary snuff | Medication (Diabetes insipidus) |
| Sauna worker's lung | Aureobasidium, Graphium spp | Contaminated sauna water |
| Sequoiosis | Aureobasidium, Graphium spp | Redwood bark, sawdust |
| Streptomyces HP | Streptomyces albus | Contaminated fertilizer |
| Suberosis | Penicillium glabrum (formerly known as Penicillium frequentans) | Moldy cork dust |
| Tap water HP | Unknown | Contaminated tap water |
| Thatched roof disease | Saccharomonospora viridis | Dried grass |
| Tobacco worker's lung | Aspergillus spp | Moldy tobacco |
| Trombone Player's lung (Brass Player's Lung) | Mycobacterium chelonae | Various Mycobacteria inside instruments[18][19] |
| Wine-grower's lung | Botrytis cinerea mold | Moldy grapes |
| Woodworker's lung | Alternaria, Penicillium spp | Wood pulp, dust |
Of these types, Farmer's Lung and Bird-Breeder's Lung are the most common. "Studies document 8-540 cases per 100,000 persons per year for farmers and 6000-21,000 cases per 100,000 persons per year for pigeon breeders. High attack rates are documented in sporadic outbreaks. Prevalence varies by region, climate, and farming practices. HP affects 0.4–7% of the farming population. Reported prevalence among bird fanciers is estimated to be 20-20,000 cases per 100,000 persons at risk."[1]
Imaging
No single imaging finding is singularly definitive of a diagnosis rather clinicians rely on a constellation of findings. Both chest radiographs and high resolution CT scans can be normal.[1][12]
Chest Radiographs
Acute presentation may reveal poorly defined a micro-nodular interstitial pattern and ground-glass opacities in the lower and mid lung zones. In addition to this, subacute presentations may show reticular nodular opacities in mid-to-lower lung zones.[1] Chronic forms may show fibrotic changes and appear like Idiopathic pulmonary fibrosis.[3]
High-Resolution Computed Tomography
This has become a common diagnostic imaging for the diagnosis and is the modality used in the Official ATS/JRS/ALAT Clinical Practice Guideline.[12] Two forms of hypersensitivity pneumonitis are fibrotic and non-fibrotic findings on chest CT. The two differ in terms of their diagnostic work up and management although there is overall between them. The non-fibrotic form is typically characterized by ground glass opacities, mosaic attenuation, ill-defined centrilobular nodules (<5 mm), and air trapping.[12] The fibrotic form is typically characterized by irregular linear opacities/coarse reticulations, traction bronchiectasis, and honeycombing, patchy ground-glass attenuation, centrilobular nodules, and mosaic attenuation.[12][20] Three-density pattern (head cheese sign) is radiological sign which shows a region of the lung with three or more different types of attenuation which can be typical for the fibrotic type.[12]
Histopathology
The acute form can be characterized by poorly formed noncaseating interstitial granulomas and mononuclear cell infiltration in a peribronchial distribution with prominent giant cells.[1] The subacute, or intermittent, form produces more well-formed noncaseating granulomas, bronchiolitis with or without organizing pneumonia, and interstitial fibrosis.[1] Much like the pathogenesis of idiopathic pulmonary fibrosis (IPF), chronic HP is related to increased expression of Fas antigen and Fas ligand, leading to increased epithelial apoptosis activation in the alveoli.[21] Cholesterol clefts or asteroid bodies are present within or outside granulomas.[1]
Pulmonary Function Testing
Pulmonary function tests (PFTs) can generally reveal a restrictive pattern [1] however, either a restrictive or obstructive pattern (or both) may emerge on PFTs. PFTs, therefore, are less useful in making a diagnosis but rather tracking improvement or deterioration in lung function following removal or addition of suspected antigens.[3] They may also demonstrate reduced diffusion capacity of lungs for carbon monoxide (DLCO).
Bronchoscopy
Bronchoalveolar lavage (BAL) is a reliable way to detect inflammation in the lung airways. Fluid analysis from the lavage extracted from the airways on bronchoscopy often reveals a total elevation in cell count in addition to an elevation in the percentage of T lymphocytes. This is a good way to help exclude other similar lung diseases like sarcoidosis, infection and Idiopathic pulmonary fibrosis.[8]
Lung biopsy
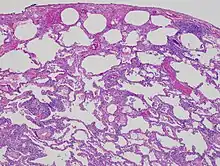
Lung biopsies can be diagnostic in cases of chronic hypersensitivity pneumonitis, or may help to suggest the diagnosis and trigger or intensify the search for an allergen. The main feature of chronic hypersensitivity pneumonitis on lung biopsies is expansion of the interstitium by lymphocytes accompanied by an occasional multinucleated giant cell or loose granuloma.[7][22]
When fibrosis develops in chronic hypersensitivity pneumonitis, the differential diagnosis in lung biopsies includes the idiopathic interstitial pneumonias.[23] This group of diseases includes usual interstitial pneumonia, non-specific interstitial pneumonia and cryptogenic organizing pneumonia, among others.[7][22]
The prognosis of some idiopathic interstitial pneumonias, e.g. idiopathic usual interstitial pneumonia (i.e. idiopathic pulmonary fibrosis), are very poor and the treatments of little help. This contrasts the prognosis (and treatment) for hypersensitivity pneumonitis, which is generally fairly good if the allergen is identified and exposures to it significantly reduced or eliminated. Thus, a lung biopsy, in some cases, may make a decisive difference.
Serum Precipitins
Assays for serum IgGs can aid in identifying possible antigenic exposures and are used as markers of exposure [12][10] However, there use is limited in making a definitive diagnosis because serum antibody levels are often elevated in those people who are exposed to an antigen but do not have the disease.[12][3][1] Up to 90% of people exposed to the antigen have precipitins but only 50% of similarly exposed people who are asymptomatic have the same precipitins.[10] False negatives are often common with serum precipitins because of lack of testing reagents for many antigens.[3]
Precipitating IgG antibodies against fungal or avian antigens can be detected in the laboratory using the traditional Ouchterlony immunodiffusion method wherein 'precipitin' lines form on agar plate. The ImmunoCAP technology has replaced this time-consuming, labor-intensive method with their automated CAP assays and FEIA (Fluorescence enzyme immunoassay) that can detect IgG antibodies against Aspergillus fumigatus (Farmer's lung or for ABPA) or avian antigens (Bird Fancier's Lung).[24]
Differential Diagnosis
Organic dust toxic syndrome presents similarly with fevers, chills a few hours after exposure to bioaerosols with toxins from fungi, however this is not a true hypersensitivity reaction because it occurs on initial exposure without a preceding sensitization [1]
In chronic disease, HP must be differentiated from very similarly presenting idiopathic pulmonary fibrosis.[12]
Although overlapping in many cases, hypersensitivity pneumonitis may be distinguished from occupational asthma in that it is not restricted to only occupational exposure, and that asthma generally is classified as a type I hypersensitivity.[25][26] Unlike asthma, hypersensitivity pneumonitis targets lung alveoli rather than bronchi.[11]
Similarly, sarcoidosis has noncaseating granuloma formation, however hilar adenopathy is often seen on chest radiographs.[12]
Treatment
The best treatment is to avoid the provoking allergen, as chronic exposure can cause permanent damage and acute disease is often self-limiting. The identification of the provoking antigen and its location must be ascertained by conducting an exposure assessment. Home cleaning is one method of antigen avoidance.[14] If avoidance is not possible, such as in the case of some workplace exposures, minimizing exposure can be accomplished through various means including the implementation of PPE or proper ventilation of a workplace.[3] Corticosteroids such as prednisolone may help to control symptoms but may produce side-effects.[27] In the case of severe, end-stage pulmonary fibrosis arising from chronic exposure, lung transplant may be the only viable option.[3] In addition to steroids for fibrotic disease, other immunosuppressants (Azathioprine, Mycophenolic acid) and anti-fibrotic agents (Nintedanib) may be used although their effectiveness is unclear[2]
Prognosis
There are few studies examining longitudinal outcomes in patients diagnosed with hypersensitivity pneumonitis. One study in the US showed about a 0.09 to 0.29 per million increase in mortality rates although the cause specific cause was unclear.[8] Most of the outcomes collected are from patients diagnosed with farmer's or bird breeder's lung and thus the degree to which this data can be extrapolated to other types of HP is uncertain. Generally outcomes for HP in those with acute disease are very good if exposure is avoided. However, those with subacute or chronic type, especially with biopsy proven fibrosis fare much poorer death rates comparable to people diagnosed with Interstitial pulmonary fibrosis.[8]
Additional images
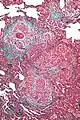 High magnification micrograph of hypersensitivity pneumonitis showing granulomatous inflammation. Trichrome stain.
High magnification micrograph of hypersensitivity pneumonitis showing granulomatous inflammation. Trichrome stain.
See also
References
- 1 2 3 4 5 6 7 8 9 10 11 12 "Hypersensitivity Pneumonitis". EMedicine. June 2006.
- 1 2 3 4 5 "Hypersensitivity pneumonitis". Nature Reviews Disease Primers. 6 (1): 66. December 2020. doi:10.1038/s41572-020-0207-8. ISSN 2056-676X. PMID 32764556. S2CID 39174418.
- 1 2 3 4 5 6 7 8 9 10 11 12 13 14 Jameson JL, Kasper DL, Longo DL, Fauci AS, Hauser SL, Loscalzo J (2018). Harrison's principles of internal medicine (20th ed.). New York. ISBN 978-1-259-64403-0. OCLC 1029074059.
{{cite book}}: CS1 maint: location missing publisher (link) - 1 2 Quirce S, Vandenplas O, Campo P, Cruz MJ, de Blay F, Koschel D, et al. (June 2016). "Occupational hypersensitivity pneumonitis: an EAACI position paper". Allergy. 71 (6): 765–779. doi:10.1111/all.12866. PMID 26913451.
- ↑ Ismail T, McSharry C, Boyd G (May 2006). "Extrinsic allergic alveolitis". Respirology. 11 (3): 262–268. doi:10.1111/j.1440-1843.2006.00839.x. PMID 16635083. S2CID 13460021.
- ↑ "Hypersensitivity Pneumonitis: Signs and Symptoms". The Regents of The University of California.
- 1 2 3 4 5 Mukhopadhyay, Sanjay. "Pathology of Hypersensitivity Pneumonitis", Retrieved on 3 May 2013.
- 1 2 3 4 5 Selman, Moisés; Pardo, Annie; King, Talmadge E. (2012-08-15). "Hypersensitivity Pneumonitis: Insights in Diagnosis and Pathobiology". American Journal of Respiratory and Critical Care Medicine. 186 (4): 314–324. doi:10.1164/rccm.201203-0513CI. ISSN 1073-449X. PMID 22679012.
- ↑ Mohr LC (September 2004). "Hypersensitivity pneumonitis". Current Opinion in Pulmonary Medicine. 10 (5): 401–411. doi:10.1097/01.mcp.0000135675.95674.29. PMID 15316440. S2CID 31344045.
- 1 2 3 LaDou J, Harrison R (2014). Current occupational & environmental medicine (5th ed.). New York: McGraw-Hill. ISBN 978-0-07-180815-6. OCLC 857214748.
- 1 2 Page 503 in: Mitchell, Richard Sheppard; Kumar, Vinay; Abbas, Abul K.; Fausto, Nelson (2007). Robbins Basic Pathology (8th ed.). Philadelphia: Saunders. ISBN 978-1-4160-2973-1.
- 1 2 3 4 5 6 7 8 9 10 11 12 13 Raghu G, Remy-Jardin M, Ryerson CJ, Myers JL, Kreuter M, Vasakova M, et al. (August 2020). "Diagnosis of Hypersensitivity Pneumonitis in Adults. An Official ATS/JRS/ALAT Clinical Practice Guideline". American Journal of Respiratory and Critical Care Medicine. 202 (3): e36–e69. doi:10.1164/rccm.202005-2032ST. PMC 7397797. PMID 32706311.
- ↑ Petnak, Tananchai; Moua, Teng (July 2020). "Exposure assessment in hypersensitivity pneumonitis: a comprehensive review and proposed screening questionnaire". ERJ Open Research. 6 (3): 00230–2020. doi:10.1183/23120541.00230-2020. ISSN 2312-0541. PMC 7520171. PMID 33015147.
- 1 2 3 4 Johannson, Kerri A.; Barnes, Hayley; Bellanger, Anne-Pauline; Dalphin, Jean-Charles; Fernández Pérez, Evans R.; Flaherty, Kevin R.; Huang, Yuh-Chin T.; Jones, Kirk D.; Kawano-Dourado, Leticia; Kennedy, Kevin; Millerick-May, Melissa (2020-12-01). "Exposure Assessment Tools for Hypersensitivity Pneumonitis. An Official American Thoracic Society Workshop Report". Annals of the American Thoracic Society. 17 (12): 1501–1509. doi:10.1513/AnnalsATS.202008-942ST. ISSN 2329-6933. PMC 7706597. PMID 33258669.
- ↑ Enelow RI (2008). Fishman's Pulmonary Diseases and Disorders (4th ed.). McGraw-Hill. pp. 1161–72. ISBN 978-0-07-145739-2.
- 1 2 3 4 5 6 7 8 9 10 11 12 Kumar 2007, Table 13-5
- ↑ Munson EL, Panko DM, Fink JG (1997). "Lycoperdonosis: Report of two cases and discussion of the disease". Clinical Microbiology Newsletter. 19 (3): 17–24. doi:10.1016/S0196-4399(97)89413-5.
- ↑ Metersky ML, Bean SB, Meyer JD, Mutambudzi M, Brown-Elliott BA, Wechsler ME, Wallace RJ (September 2010). "Trombone player's lung: a probable new cause of hypersensitivity pneumonitis". Chest. 138 (3): 754–756. doi:10.1378/chest.10-0374. PMID 20823006.
- ↑ "Sour Note: Sax Can Cause Lung Disease". ABC News. 7 September 2010. Retrieved 4 April 2018.
- ↑ Rizi, Farid Rajaee. "Hypersensitivity pneumonitis | Radiology Case | Radiopaedia.org". Radiopaedia. doi:10.53347/rid-150222. Retrieved 2022-12-01.
- ↑ Jinta T, Miyazaki Y, Kishi M, Akashi T, Takemura T, Inase N, Yoshizawa Y (October 2010). "The pathogenesis of chronic hypersensitivity pneumonitis in common with idiopathic pulmonary fibrosis: expression of apoptotic markers". American Journal of Clinical Pathology. 134 (4): 613–620. doi:10.1309/AJCPK8RPQX7TQRQC. PMID 20855643.
- 1 2 Mukhopadhyay S, Gal AA (May 2010). "Granulomatous lung disease: an approach to the differential diagnosis". Archives of Pathology & Laboratory Medicine. 134 (5): 667–690. doi:10.5858/134.5.667. PMID 20441499.
- ↑ Ohtani Y, Saiki S, Kitaichi M, Usui Y, Inase N, Costabel U, Yoshizawa Y (August 2005). "Chronic bird fancier's lung: histopathological and clinical correlation. An application of the 2002 ATS/ERS consensus classification of the idiopathic interstitial pneumonias". Thorax. 60 (8): 665–671. doi:10.1136/thx.2004.027326. PMC 1747497. PMID 16061708.
- ↑ Khan S, Ramasubban S, Maity CK (1 July 2012). "Making the case for using the Aspergillus immunoglobulin G enzyme linked immunoassay than the precipitin test in the diagnosis of allergic bronchopulmonary aspergillosis". Indian Journal of Allergy, Asthma and Immunology. 26 (2): 89. doi:10.4103/0972-6691.112555. Retrieved 4 April 2018.
- ↑ "Lecture 14: Hypersensitivity". Archived from the original on 2006-02-06. Retrieved 2008-09-18.
- ↑ "Allergy & Asthma Disease Management Center: Ask the Expert". Archived from the original on 2007-02-16. Retrieved 2008-09-18.
- ↑ "Hypersensitivity Pneumonitis Treatment - Conditions & Treatments - UCSF Medical Center". www.ucsfhealth.org. Retrieved 4 April 2018.