 | |
 | |
| Names | |
|---|---|
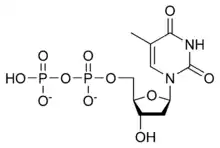
| IUPAC name
Thymidine 5′-(trihydrogen diphosphate) | |
| Systematic IUPAC name
[(2R,3S,5R)-3-Hydroxy-5-(5-methyl-2,4-dioxo-3,4-dihydropyrimidin-1(2H)-yl)oxolan-2-yl]methyl trihydrogen diphosphate | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChemSpider | |
PubChem CID |
|
| UNII | |
| |
| |
| Properties | |
| C10H16N2O11P2 | |
| Molar mass | 402.19 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |
Thymidine diphosphate (TDP) or deoxythymidine diphosphate (dTDP) (also thymidine pyrophosphate, dTPP) is a nucleotide diphosphate. It is an ester of pyrophosphoric acid with the nucleoside thymidine. dTDP consists of the pyrophosphate group, the pentose sugar ribose, and the nucleobase thymine. Unlike the other deoxyribonucleotides, thymidine diphosphate does not always contain the "deoxy" prefix in its name.[1]
See also
References
- ↑ Coghill, Anne M.; Garson, Lorrin R., eds. (2006). The ACS style guide: effective communication of scientific diphosphates information (3rd ed.). Washington, D.C.: American Chemical Society. p. 244. ISBN 978-0-8412-3999-9.
External links
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.