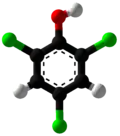
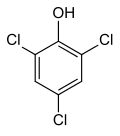
 | |||
| |||
 | |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
2,4,6-Trichlorophenol | |||
| Identifiers | |||
3D model (JSmol) |
|||
| 776729 | |||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| ECHA InfoCard | 100.001.633 | ||
| EC Number |
| ||
| 3766 | |||
| KEGG | |||
PubChem CID |
|||
| RTECS number |
| ||
| UNII | |||
| UN number | 2020 | ||
CompTox Dashboard (EPA) |
|||
| |||
| |||
| Properties | |||
| C6H2Cl3OH/C6H3Cl3O | |||
| Molar mass | 197.45 g/mol | ||
| Appearance | yellow-whitish lumps or powder | ||
| Density | 1.4901 g/cm3 at 75 °C[1] | ||
| Melting point | 69.5 °C (157.1 °F; 342.6 K)[1] | ||
| Boiling point | 249 °C (480 °F; 522 K)[1] | ||
| 0.069 g/100 g H2O[2] | |||
| Hazards | |||
| GHS labelling: | |||
   | |||
| Warning | |||
| H302, H315, H319, H351, H410 | |||
| P201, P202, P264, P270, P273, P280, P281, P301+P312, P302+P352, P305+P351+P338, P308+P313, P321, P330, P332+P313, P337+P313, P362, P391, P405, P501 | |||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references | |||
2,4,6-Trichlorophenol, also known as TCP, phenaclor, Dowicide 2S, Dowcide 2S, omal, is a chlorinated phenol that has been used as a fungicide, herbicide, insecticide, antiseptic,[3] defoliant, and glue preservative.[4] It is a clear to yellowish crystalline solid with a strong, phenolic odor. It decomposes on heating to produce toxic and corrosive fumes including hydrogen chloride and chlorine.
Preparation
2,4,6-Trichlorophenol is produced industrially by the electrophilic chlorination of phenol:[5]
Health effects
In animal models, consumption of 2,4,6-trichlorophenol leads to an increased incidence of lymphomas, leukemia, and liver cancer.[6][7] It is classified as Group B2 (probable human carcinogen) by the United States Environmental Protection Agency.[7] The technical grade of this substance may contain polychlorinated dibenzodioxins (PCDDs), polychlorinated dibenzofurans (PCDFs), and other contaminants.[8]
Environmental effects
2,4,6-Trichlorophenol is an environmental pollutant that has been found in fresh water lakes such as the Great Lakes.[9]
See also
- Trichlorophenol (for other isomers).
References
- 1 2 3 Haynes, p. 3.522
- ↑ CRC handbook of chemistry and physics : a ready-reference book of chemical and physical data. William M. Haynes, David R. Lide, Thomas J. Bruno (2016-2017, 97th ed.). Boca Raton, Florida. 2016. ISBN 978-1-4987-5428-6. OCLC 930681942.
{{cite book}}: CS1 maint: location missing publisher (link) CS1 maint: others (link) - ↑ Ogunniyi TA, Oni PO, Juba A, Asaolu SO, Kolawole DO (2000-01-05). "Disinfectants/antiseptics in the management of guinea worm ulcers in the rural areas". Acta Tropica. 74 (1): 33–38(6). doi:10.1016/S0001-706X(99)00057-1. PMID 10643905.
- ↑ "Safety data for 2,4,6-trichlorophenol". University of Oxford. 2005-09-05. Archived from the original on 2007-10-14. Retrieved 2007-11-16.
- ↑ Muller, François; Caillard, Liliane (2011-10-15), "Chlorophenols", in Wiley-VCH Verlag GmbH & Co. KGaA (ed.), Ullmann's Encyclopedia of Industrial Chemistry, Weinheim, Germany: Wiley-VCH Verlag GmbH & Co. KGaA, pp. a07_001.pub2, doi:10.1002/14356007.a07_001.pub2, ISBN 978-3-527-30673-2, retrieved 2022-03-13
- ↑ "2,4,6-Trichlorophenol". The Carcinogenic Potency Database Project, University of Berkeley. 2007-10-03. Archived from the original on 4 December 2007. Retrieved 2007-11-16.
- 1 2 "2,4,6 Trichlorophenol". United States Environmental Protection Agency. Jan 2000. Retrieved 2007-11-16.
- ↑ "2,4,6-Trichlorophenol". IPCS. Nov 1998. Archived from the original on 2013-06-27. Retrieved 2007-11-16.
- ↑ TP Halappa Gowdal; John D Lock; Ruth G Kurtz (Feb 1985). "A comprehensive study of risk assessment for a hazardous compound of public health concern". Water, Air, & Soil Pollution. 24 (2): 189. Bibcode:1985WASP...24..189H. doi:10.1007/BF00285444. S2CID 96067556.
Cited sources
- Haynes, William M., ed. (2016). CRC Handbook of Chemistry and Physics (97th ed.). CRC Press. ISBN 9781498754293.